|
Page History
| Multiexcerpt | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
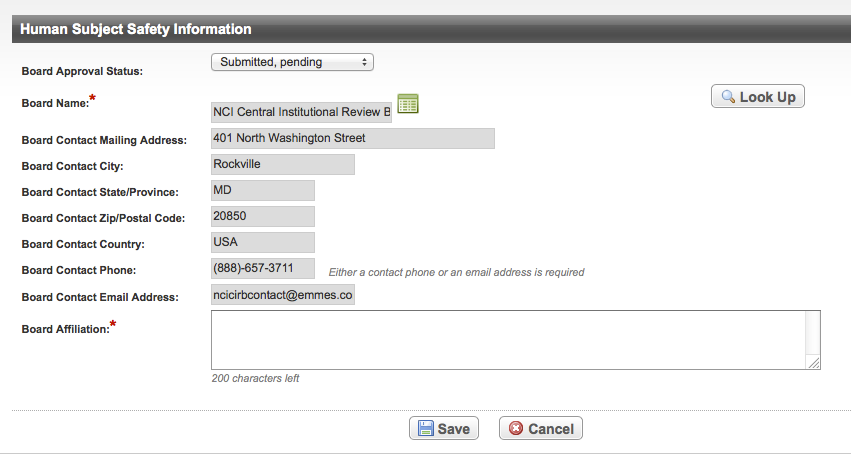
The information you are required to provide depends on the Board approval status and other indicators, as noted in the instructions that follow. Human subject safety information is optional for Abbreviated trials.
Some fields in this section are optional, and some may be required only when you have selected certain attributes in preceding fields. For example, if the Board Approval Status is "Submitted, exempt", the Board Name, Board Affiliation, and Board Contact fields are mandatory, but the Board Approval Number is optional. The following table provides a matrix of field requirement dependencies. Matrix of field requirements
How to Abstract Human Subject Safety Information
|