|
Page History
Trials can be excluded from Data Table 4 (DT4) reports at either the Trial or Family level:
- Trial: Exclude a trial from appearing on all Families DT4 report, typically used for trials that are non-oncology based.
- Family. Exclude a trial from appearing on a specific Family or Families DT4 report. Typically Expanded Access trials where a Center determines the particular trial should not be included on their DT4 report.
Search for the trial of interest. For instructions, refer to Searching for Trials in PA.
In the search results, Select the NCI Trial Identifier link for that trial. The Trial Identification page opens.
On the Trial Identification page, check out the trial. For instructions, refer to Checking In and Checking Out Trials. (This checkout step is optional for Super Abstractors.)
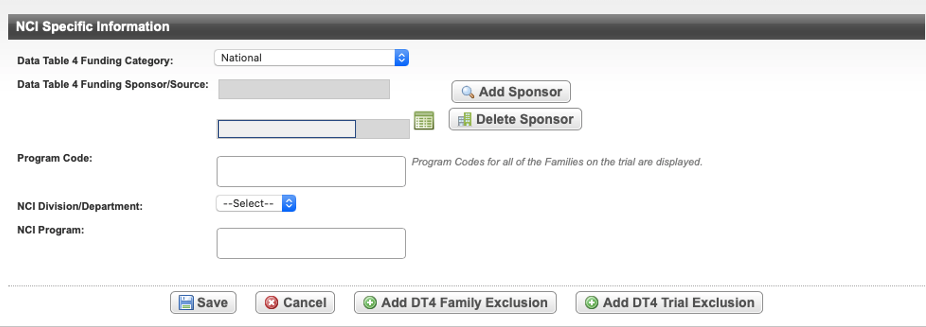
On the Administrative Data menu, click NCI Specific Information. The NCI Specific Information page opens.
Data Table 4 Family Exclusions
Adding a Family Exclusion
- On the NCI Specific Information page, select Add DT4 Family Exclusion. The Add Family for DT4 Exclusion page opens.
- In the Family drop down menu, select the desired Family.
- In the DT4 Family Exclusion Reason field, enter a reason for the exclusion.
- Select Save. The NCI Specific Information page opens.
Managing Family Exclusions
On the NCI Specific Information page for trials with existing DT4 Family Exclusions, the following options are available:
Field Label | Description/Instructions |
|---|---|
| Add DT4 Family Exclusion | Add an additional family for DT4 exclusion. |
Convert to DT4 Trial Exclusion | Change exclusion from a Family Exclusion to a Trial Exclusion |
Delete Selected DT4 Families | To remove one or more families from the DT4 exclusion. |
Data Table 4 Trial Exclusions
Adding a Trial Exclusion
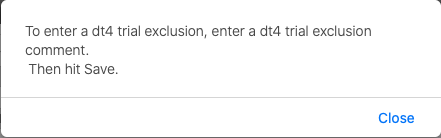
- On the NCI Specific Information page, select Add DT4 Trial Exclusion.
- Select Close on the following window
- The NCI Specific Information page re-opens with the DT4 Trial Exclusion Reason field available.
- In the DT4 Trial Exclusion Reason field, enter a reason for the exclusion.
- Select Save. A Message Recorded confirmation is displayed.
Deleting a Trial Exclusion (Can only be completed by a Super Abstractor)
- Navigate to the NCI Specific Information page for a trial with an existing DT4 Trial Exclusion. Refer to Searching for Trials in PA.
- Select Delete Exclusion.
- An Update Succeeded message will be displayed and the NCI Specific Information page will re-open.
The system processes CTEP (Cancer Therapy Evaluation Program) and DCP (Division of Cancer Prevention) accrual records submitted in batch files to the CTRP via CDUS (Clinical Data Update System). If a batch file contains accruals for a site that has not been added to a given trial in the CTRP, the system generates an email message to the submitter that includes a list of the sites for which accruals were submitted but not recorded. These sites' accrual records remain in a separate table, or queue, pending further processing.
The system re-submits and processes the batch files daily. If the system subsequently finds that a participating site has been added to the trial in the CTRP, it removes the site from the pending accruals queue.
However, if it finds that the site still has not been added in the CTRP, it notifies the submitting Super Abstractor who can then add the site to the trial in PA. If the Super Abstractor determines that the error-producing participating site is invalid, the site and its accrual records can be deleted from the queue.
| Info |
|---|
The Pending Accruals feature is available to Super Abstractors only. |
- On the main menu, click Pending Accruals Queue.
The Pending Accruals Queue page appears. - To search for accrual records for a given trial, enter the trial's Identifier in the field provided, and then click Search.
- To delete a record, select the appropriate check box in the Delete column, and then click Remove Site From Queue.