|
Page History
This page provides an overview of the changes in CTRP. For any questions or issues regarding these feature enhancements, please contact the CTRO (NCICTRO@mail.nih.gov).
May 2024
New CTRP trials.nci.nih.gov login page and new Accrual application are now live.
New Login page with accompanying landing page allows users to select and launch the new Accrual, Registration, Protocol Abstraction, or Person/Organization CTRP applications.
New Accrual: CTRP has developed a new Accrual Reporting application available via trials.nci.nih.gov. The new version maintains most of the previous Accrual (e.g., now referred to as Classic Accrual)'s functionality; however, the user interface is updated, and user experience is enhanced. These enhancements include:
- Trial Search
- The Accrual Counts and Trial Search page in classic Accrual have been combined. Please note the Accrual Counts page is decommissioned.
- The ability to show/hide columns is expanded to the newly added fields absorbed from the Accrual Counts page.
- Search and filter capabilities have been expanded beyond NCI Trial Identifier, ClinicalTrials.gov Identifier, and Official Title fields.
- The ability to filter prior submissions by the NCI Trial Identifier is available under the “Actions” column.
- Study Subject
- Study subject records still inherit the trial record’s selected accrual disease terminology; however, study subject records are now allowed to not match the accrual disease terminology selected at trial-level.
- The field “Study Subject Gender” is relabeled to display “Study Subject Sex” throughout the application.
- The “Method of Payment” and “Registering Group Identifier” fields have been decommissioned throughout the new Accrual application.
- Study Subject ZIP Code will allow users to enter 9 digits for US-based subjects; however, new Accrual will only save the first 5 digits.
- Prior Submissions
- The “Rejection Reason” column has been added to the page and is included in file export up to a 1,000 characters.
| Tip | ||
|---|---|---|
| ||
The generated email notification for accrual submission is still providing the most comprehensive information, especially for large batch submissions. The list displayed in the new Accrual application can provide initial, aggregated information to users. |
- Session Time-out Changes
- The new application no longer has an automatic log-out after sixty-minutes.
Learn how to use the new Accrual via updated CTRP Accrual User Guide
- Logging in to CTRP page
- Accrual User Guide (updated with new Accrual information)
- Classic Accrual User Guide.pdf (right click to open in new tab)
Please note: The new Accrual application is available alongside the Classic Accrual application. Users will have the option to utilize either application for a brief period, until the Classic Accrual application is deprecated and no longer available. The use of the new Accrual application is highly encouraged.
| Tip | ||
|---|---|---|
| ||
Some users may need to clear their browser cache in order to access the New Accrual reporting application. |
To access Classic Accrual: On main landing page, within the top right-hand menu, a user can click the Profile down button, and the Classic Accrual application is listed within the Quick Links menu.
Please report any technical questions about CTRP applications using ctrp_support@nih.gov.
March 2023
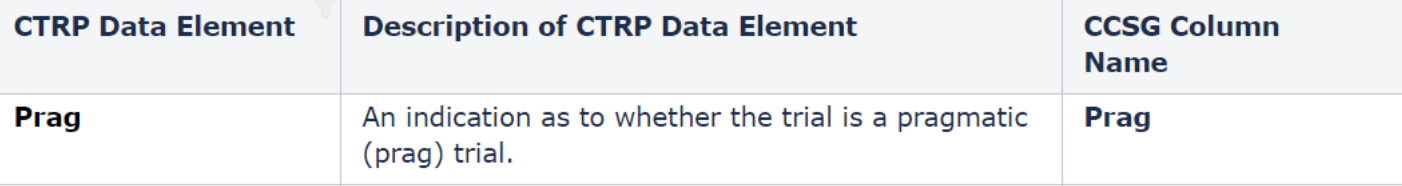
Update to the CTRP-generated DT4 report display to include the pragmatic trial indicator. The field is labeled ‘Prag’ with values of ‘Y’ or ‘N’ and is abstracted by the CTRO. This value will be included in any format of the CTRP-generated DT4 report (e.g., pdf, csv). This update does not have any impact on CTRP REST Services or CTRP REST Service users.
Prag values:
Y – Yes, this is a pragmatic trial
...
Update to the CTRP Trial Summary Report (TSR) to include the pragmatic trial indicator. The field is labeled “Pragmatic Trial” and is in the ‘Trial Design’ table.
“Pragmatic Trial” values on the TSR:
Yes – This is a pragmatic trial
No – This is not a pragmatic trial
No Data Available – CTRP currently does not have information as to whether or not this is a pragmatic trial
...
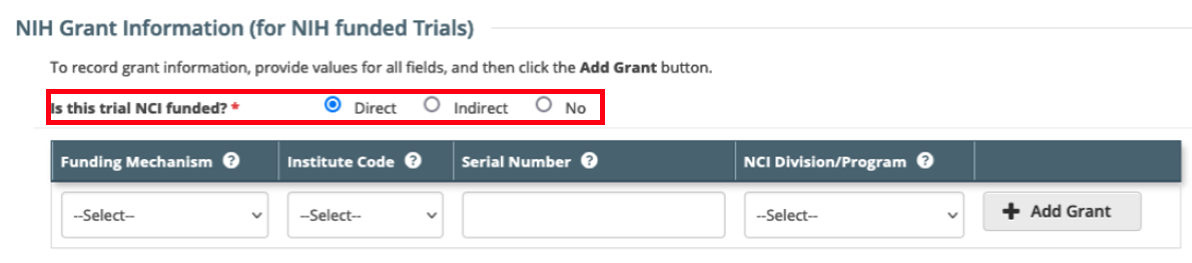
Update to the NIH Grant Information section in the CTRP Registration application. The field 'Is this trial funded by an NCI Grant?' with values of Yes/No has been updated to 'Is this trial NCI funded?' with values of Direct/Indirect/No. This field is available through the web application when registering and amending trials through the CTRP Registration user interface. This update does not have any impact on CTRP REST Services or CTRP REST Service users.
Definitions:
...
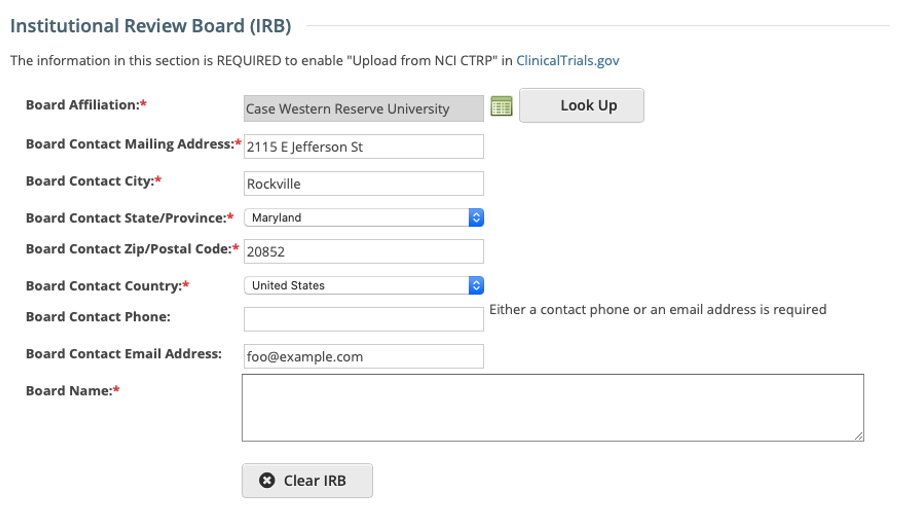
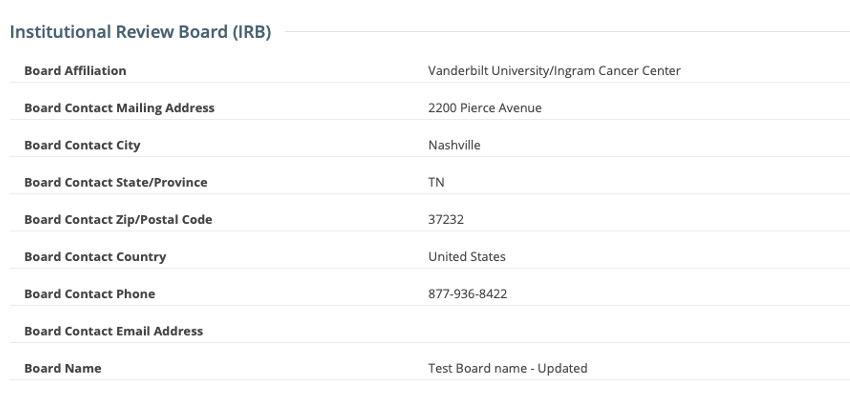
Registering, Amending or Updating/Verifying a non-industrial trial: IRB fields are modifiable
Viewing a trial on the Trial Search page: IRB fields are read-only
For additional information about the IRB fields in the CTRP Registration application, please visit these CTRP User Guide pages:
...
- Okta Verify: Use a 'Push Notification' sent to the Okta app on the users mobile device.
- SMS Authentication: Enter a single-use code sent to the users mobile device.
- Voice Call Authentication - Phone call authentication by following voice instructions.
New CTRP login page
For additional information about creating and setting up an Okta account, please visit these CTRP User Guide pages:
...