There are multiple ways to access the Trial Identification page, including the following:
- Search for a trial using the Search Trial feature. Refer to Searching for Trials in PA. In the Search Results list, click the NCI Trial Identifier link for the clinical trial you want to abstract.
- or - - On the abstraction dashboard Workload or Results tab, click the NCI Trial Identifier link for the clinical trial you want to abstract. On the main menu, click Trial Identification.
The Trial Identification page displays information about the trial as it was registered in the CTRP system:
The top of the Trial Identification page contains pertinent aspects of the trial, including lead organization, principal investigator, and trial submitter. This summary remains visible at the top of each page as you progress through the trial validation and abstraction.
To view the principal investigator’s or trial submitter’s contact information, click the appropriate link in the summary. A pop-up window displays the information.
- The main section of the Trial Identification page displays most of the same information as the summary in addition to other trial identifiers (including duplicate and obsolete identifiers), trial category, and trial type. The combination of these elements uniquely defines the trial in the CTRP.
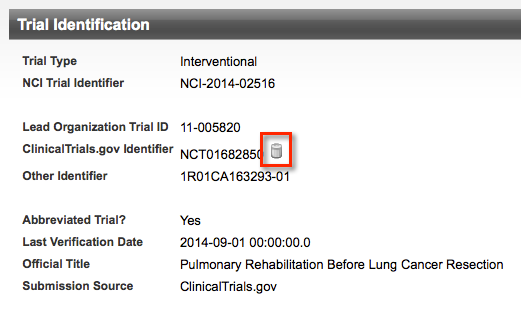
For rejected trials, the system displays a Delete icon next to the NCT ID in the Trial Identification section.
To delete the NCT ID from the trial, click the Delete icon, and then confirm that you want to delete the ID.
Other features on this page enable you to do the following:
- Check trials in/out. See Checking In and Checking Out Trials .
- Assign trials to other abstractors (Super Abstractors only). See Assigning Trials to Abstractors.
- Assign priority levels (Super Abstractors only). See Assigning Trial Priorities .