You can add one or more study subject accrual records for any trial to which you have been granted access.
1
See Requesting Permission to Submit Accrual Data .
Study subject records include demographic data as well as the disease name.
How to Add Study Subject Records
- Select the trial you want to work with by following instructions in Selecting Listed Trials in Accrual or Searching for Trials in Accrual , and clicking the corresponding NCI Trial Identifier link.
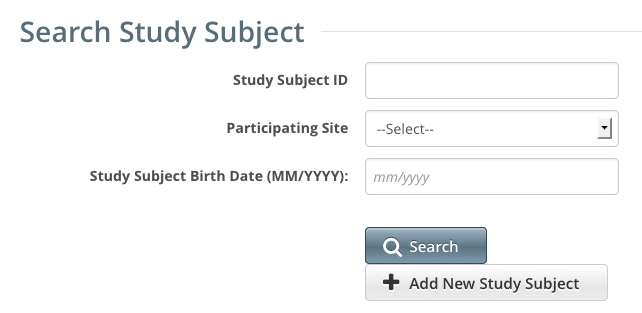
The Search Study Subject page appears.
- Click Add New Study Subject.
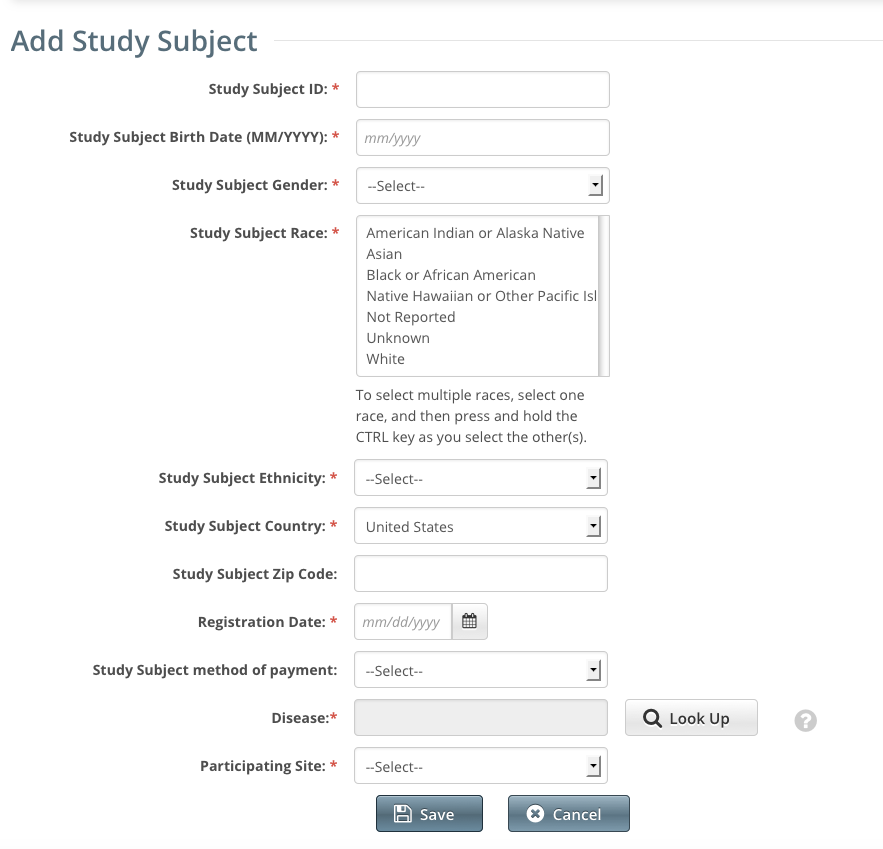
The Add Study Subject page appears.
Enter the appropriate demographic information in the text fields and drop-down lists. The following table describes the fields. An asterisk (*) indicates a required field. For a list of valid values and formats for each field, see Accrual Data Elements for Complete Trials .
Descriptions and instructions for study subject demographic data fieldsStudy Subject Information
Instruction/Description
Study Subject ID*
Enter the unique Patient ID as per the lead organization or the study site where the subject is registered.
Study Subject Birth Date*
Enter the subject's month and year of birth in the format
MM/YYYY.Study Subject Gender*
Select the subject’s gender. If gender information is not available, select Unknown.
Study Subject Race*
Select one or more values for race.
To select multiple races, select one race, and then press and hold the CTRL/CMD key as you select the other(s).
Study Subject Ethnicity*
Select a value for ethnicity.
Study Subject Country*
Select the subject’s country of origin.
Study Subject Zip Code
Enter the subject’s Zip Code if known. This field is mandatory if the country of origin is United States.
Registration Date*
Enter the date that the subject was registered for the trial.
Study Subject Method of Payment
For United States study subjects only, select the appropriate payment method.
Site*
Click Look Up, and follow the instructions in Selecting Sites for Study Subject Records Using ICD-O-3 Codes.
Mandatory for ICD-O-3 trials unless you record a CD-O-3 Disease Code. Optionally, record both Site and Disease codes.
Site codes are available at http://training.seer.cancer.gov/head-neck/abstract-code-stage/codes.html.Disease*
Click Look Up, and follow the instructions in Selecting Diseases for Study Subject Records.
Mandatory for ICD-9 and SDC trials; and for ICD-O-3 trials unless you record a CD-O-3 Site Code.
Use Histology codes when C codes are not available (for example, for Multiple Myeloma, NHL, Leukemia).
Participating Site*
Select the appropriate site from the drop-down list.
- Click Save.
The study subject record appears in the List of Study Subjects.
You can submit accrual data to the NCI CTEP's Clinical Data Update System (CDUS) on a quarterly basis (i.e., December 31, March 31, June 30) up to 30 days following the end of the quarter.
For Complete trials, lead organizations report all subjects accrued for the trial (both in the lead organization and in all participating sites). For Abbreviated trials, each participating site reports the number of its own accruals (accrual count) only.
---
- If your organization currently submits subject accrual information for studies to CTEP or DCP via the CDUS system, continue to report subject accrual information via CDUS. The NCI will manage the transfer of subject accrual data for CDUS trials internally. Otherwise, submit your organization's subject accrual data to CTRP. ↩