How to Amend Trials
- On the toolbar, click Search > Clinical Trials.
The Search Trials page appears. - Click Search > My Trials.
The Search Results page displays the results of your search and actions available (if any) for each record.
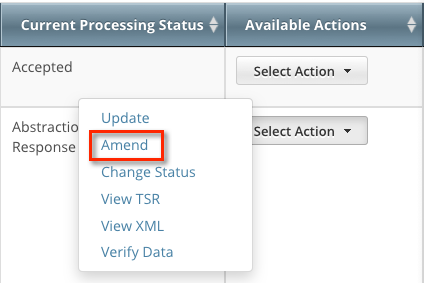
In the Available Actions column, click Select action > Amend. The Amendment Trial page displays the data currently registered with the CTRP.
In the Amendment Details section, select or enter the appropriate information in the drop-down lists and text fields. The following table describes the fields. An asterisk (*) indicates a required field.
Field Label Description/Instructions Amendment Number
Enter an appropriate number.
Amendment Date* Select or enter an appropriate date. Select or enter the appropriate information in the remaining text fields and drop-down lists, following the instructions provided in Registering New Trials .
You are required to provide information for all fields marked with an asterisk (*).
You can select a different disease code only if the trial has not accrued any subjects to date.
- Review the amendment. See Reviewing and Submitting Trial Amendments .
- Submit the amended trial to the CTRP.
The system sends you an email notification — with the details of what has changed — whenever you amend accepted trials.
A trial can accumulate program codes from different organization families. For example, a participating site might belong to a different organization family than the lead organization. When you amend a trial, the Program Code field displays all codes from the master list for the organization family of the lead organization.