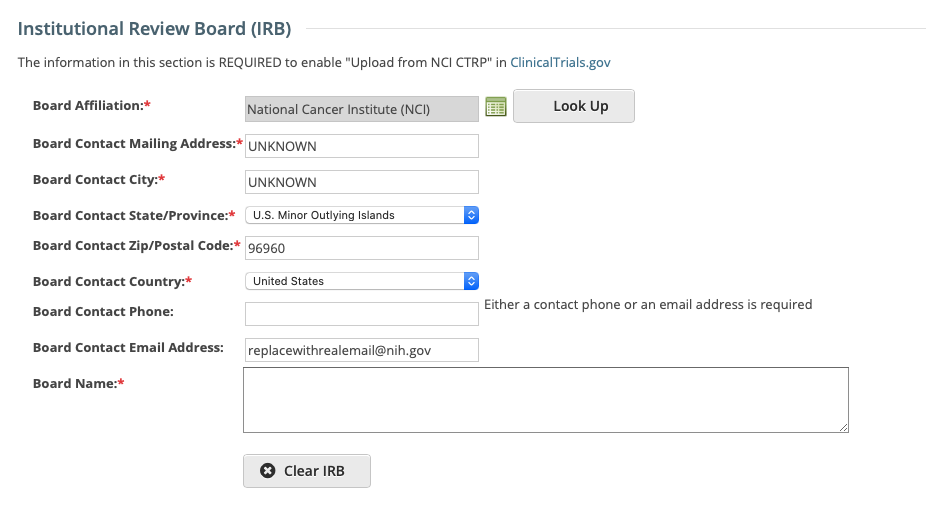
The Institutional Review Board (IRB) section is optional, however there are required fields if completing this section which are indicated with an asterisk below.
How to complete the Institutional Review Board (IRB) section
1. Select or enter the appropriate information in the text fields and drop-down lists. The following table describes the fields.
Field Label | Description/Instructions |
|---|---|
Board Approval Number* | If the trial has been submitted and approved, type the number assigned to the protocol by the human subjects review board in the field. If the number does not appear in the IRB approval document, abstract the IRB approval date from the IRB document in the following format: mm/dd/yyyy. The IRB approval date may be labeled as Action Date or Approval Date. |
Board Affiliation* | Click Look Up to search for and select the Board name. If the name is not listed, register it with the CTRO. See Searching for Organizations. |
Board Name | The name of the affiliated organization. |