Date
Attendees
Goals
- Review, discuss and prioritize ICDC study submission proposals
Previous Action items
- Musk, Philip (NIH/NCI) [C]to reach out to Roel to communicate metadata fields needed for existing publication
- Kuffel, Gina (NIH/NCI) [C] to put RNA-Seq broad normalization on the agenda for next time.
Discussion items
| Item | Who | Notes |
|---|---|---|
| New UBC study | Kuffel, Gina (NIH/NCI) [C] |
|
| Study Updates |
| |
| Feature Updates |
| |
| Material Transfer Agreement (MTA) | Kuffel, Gina (NIH/NCI) [C] |
|
| RNA-Seq broad normalization |
|
Minutes (Not Verbatim)
PM- We have the green light from Shaying after they had a courtesy preview of the data on our QA env. Testing is in progress to ensure nothing breaks in our UI. This is now in our hands waiting for a production release.
PM- Use the courtesy preview as an opportunity to reveal features in development.
PM- tumor_sample_origin captures the nature of the tumor (metastatic for example). Sample site is concatenated with this field to capture additional information. New sample profile visualizations will further capture this as well.
PM- 2 studies for possible submission, coming out of the University of Florida, data submitter is aware of submission process.
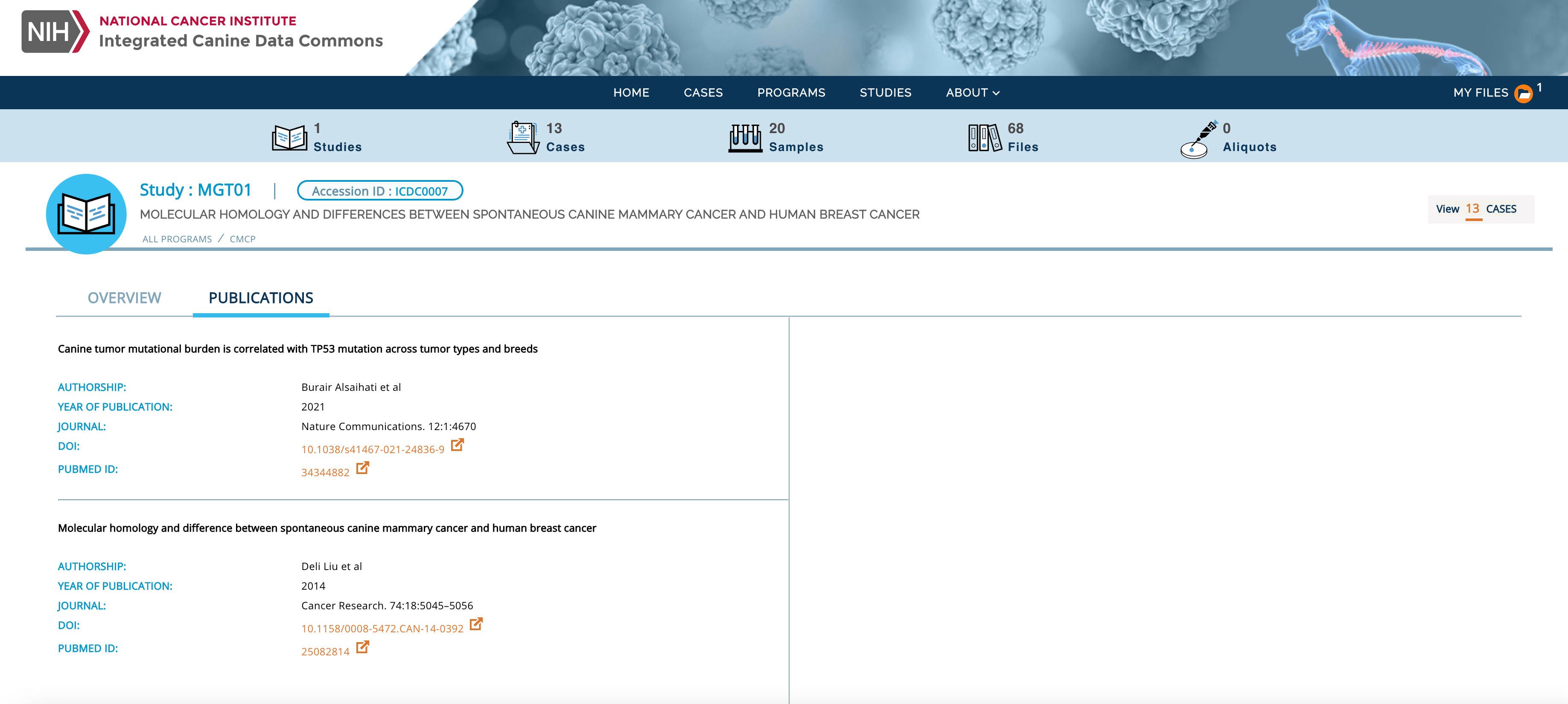
Figure 1: New publications tab to capture study level metadata.
Action items
- Kuffel, Gina (NIH/NCI) [C] to send Doodle for a new meeting time.
- Kuffel, Gina (NIH/NCI) [C] to send an email to Shaying that contains submission request form for 15 TB study.
- Pihl, Todd (NIH/NCI) [C] to put together an email for Milner