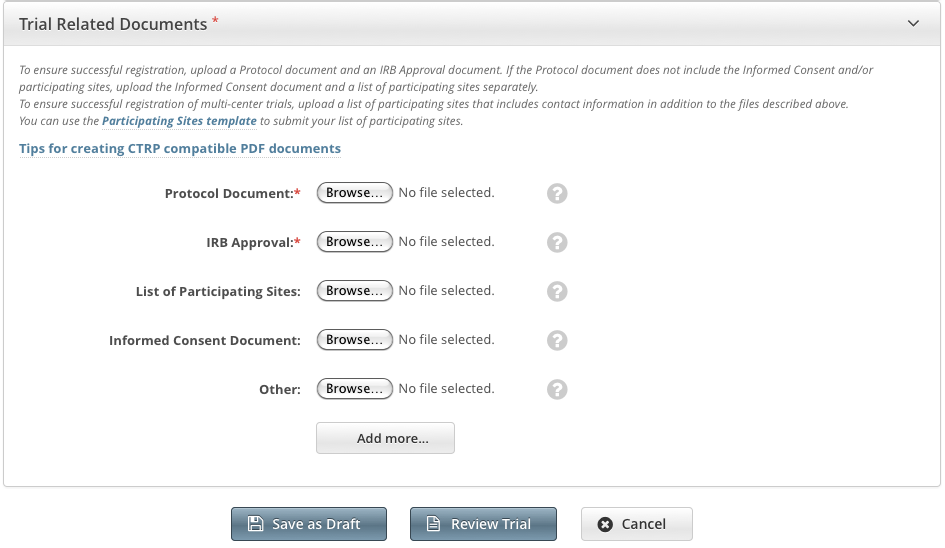
When registering Complete trials, you must upload the following types of documents:
Informed Consent (if not included in the protocol document).
Currently, the system requires you to supply each of your documents as one of the following formats:
How to Submit Trial Related Documents
Follow your browser/operating system instructions to navigate to, select, and open the appropriate document.
Repeat the steps above for each type of document.
You can upload more than one (1) "Other" document. After you have uploaded the first of your "Other" documents, click the Add More link. The system displays a new Other document field. |