On the Trial Identification page, check out the trial. For instructions, refer to Checking In and Checking Out Trials. (This checkout step is optional for Super Abstractors.)
On the Validation menu, click Trial Related Documents.
The Trial Related Documents page appears.
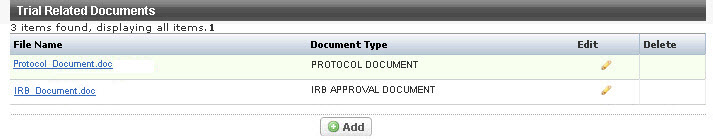
You must submit a participating sites document for a multi-site trial if the participating sites data are not included in the protocol document. |
- To add a new document, click Add and follow the instructions provided in Adding Documents.
- To modify or replace an existing document, click the Edit icon and follow the instructions provided in Editing and Replacing Documents.
To delete a document other than a Protocol or IRB Approval document, click the Delete icon in the Delete column and follow the instructions provided in Deleting Documents.
You can add, edit, or delete only those documents that are not required. You only can replace the required ones.