- In the list of trials, click the NCI trial identifier link for the trial you want to edit. A page appears with several sections, including the Final Record Clean-up & Release section.
- In the panel next to the pie chart, under Results Cover Sheet, type the trial ID and click Search. The Cover Sheet page appears with multiple sections, including the Final Record Clean-up & Release section.
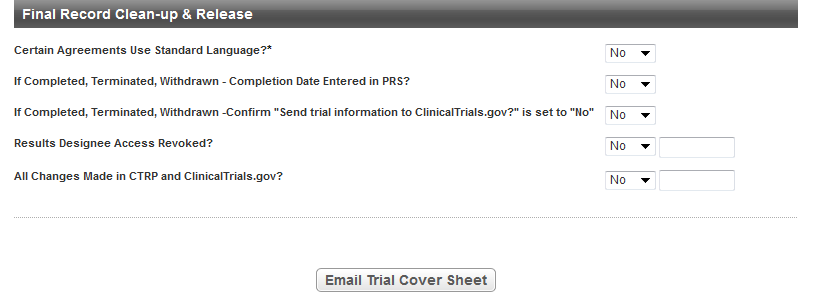
In the Final Record Clean-up & Release section, enter the appropriate information in the fields. The following table describes the fields. An asterisk (*) indicates a required field.
| Field | Instructions | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Certain Agreements Use Standard Language?* | Select Yes in this field after you have provided the following answers in PRS:
| ||||||||
If Completed, Terminated, Withdrawn – Completion Date Entered in PRS? | This field becomes relevant when the trial has a Current Trial Status of Complete, Administratively Complete, or Withdrawn in the PA application. In that case, select Yes in this field if the following is true: In the PA application, under Trial Status, the Completion Date for the trial has a date, and PRS has the same date. | ||||||||
| If Completed, Terminated, Withdrawn – Confirm “Send trial information to ClinicalTrials.gov?” is set to “No” | This field becomes relevant when the trial has a Current Trial Status of Complete, Administratively Complete, or Withdrawn in the PA application. In that case, select Yes in this field if the following is true: In the PA application, in the NCI Specific Information section, the trial has No in the Send trial information to ClinicalTrials.gov? field. | ||||||||
| Results Designee Access Revoked? | This field becomes relevant when the PRS Team publishes the trial with results in ClinicalTrials.gov. In that case, select Yes in this field if the CTA removed the Results Designee user account from the trial record access list in PRS. If you select Yes, type or select the date on which the CTA revoked the Results Designee's access. If you type the date, use the following format: | ||||||||
| All Changes Made in CTRP and ClinicalTrials.gov? | This field becomes relevant when the CTRO makes changes to the trial since the Reporting in Process date, in any system (CTRP or PRS). In that case, select Yes in this field if both of the following conditions are true:
If you select Yes, type or select the date on which the CTA uploaded the changes. If you type the date, use the following format: |