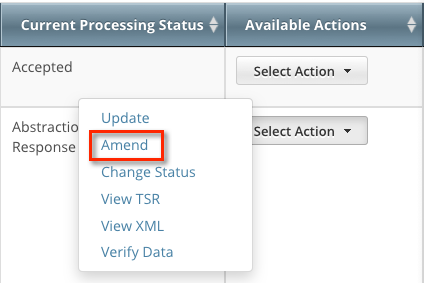
In the Available Actions column, click Select Action > Amend. The Amendment Trial page displays the data currently registered with the CTRP.
Screenshot TBD. |
Make changes to the fields as necessary. The system requires you to provide information for all fields marked with an asterisk (*). The instructions are the same for trial registration and trial amendment, with some exceptions:
In the Amendment Details section, specify the appropriate information in the various fields. The following table describes the fields.
Field Label Description/Instructions Amendment Number
Enter an appropriate number.
Amendment Date* Select or enter an appropriate date. A trial can accumulate program codes from different organization families. For example, a participating site might belong to a different organization family than the lead organization. When you amend a trial, the Program Code field displays all codes from the master list for the organization family of the lead organization.
Primary Completion Dates are optional for non-interventional trials and for DCP trials. In both cases, the system excludes such trials when submitting XML documents to ClinicalTrials.gov. Otherwise, Primary Completion Dates are required.
For instructions on recording each field otherwise, refer to Registering New Trials.