Search for the trial of interest. For instructions, refer to Searching for Trials in PA.
In the search results, click the NCI Trial Identifier link for that trial. The Trial Identification page appears.
On the Trial Identification page, check out the trial. For instructions, refer to Checking In and Checking Out Trials. (This checkout step is optional for Super Abstractors.)
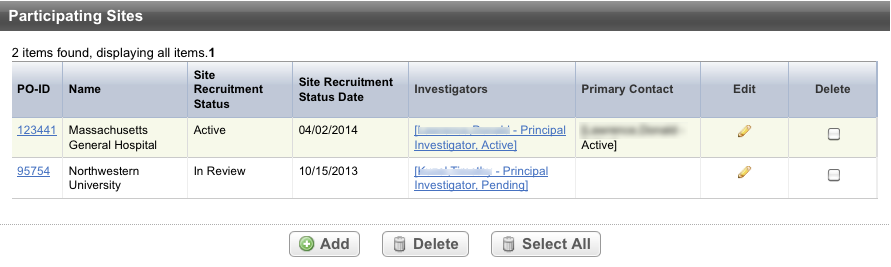
Click Add. The Participating Sites page displays three tabbed sections: Participating Site, Investigators, and Contact. The Participating Site tab is displayed by default.
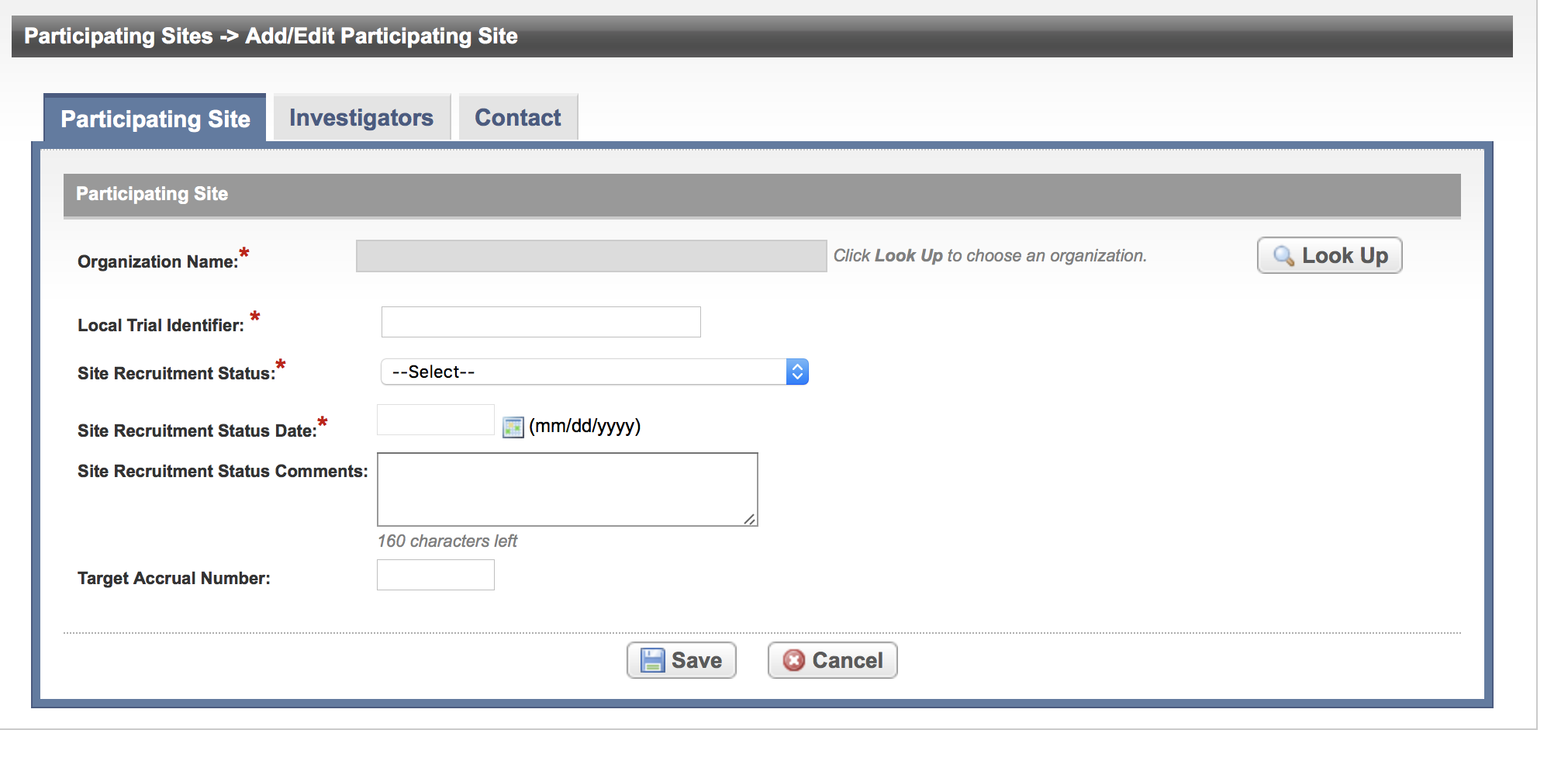
In the various fields, specify the appropriate information. The following table describes the fields. An asterisk (*) indicates a required field.
Field Label | Description/Instructions | |
|---|---|---|
Organization Name* | Click Look Up and follow the instructions in Searching for Organizations. | |
Local Trial Identifier* | Enter the site trial identifier. | |
Site Recruitment Status* |
| |
Site Recruitment Status Date | Enter the date that the status was recorded. The date must be the current date or earlier. | |
| Site Recruitment Status Comments | Enter one or more comments about the site recruitment status. | |
| Target Accrual Number | If the lead organization or participating site is a member of a Cancer Center family of organizations, enter the accrual number. |