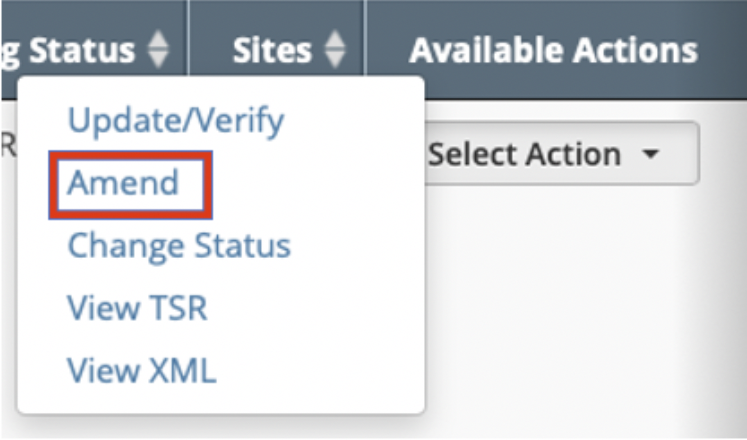
The Amend Trial page can be accessed by either of the following methods below:
Using the Trials to Verify menu
Using the Search menu
Make changes to the fields as necessary. The system requires you to provide information for all fields marked with an asterisk (*). The instructions are the same for trial registration and trial amendment, with some exceptions:
In the Amendment Details section, specify the appropriate information in the various fields. The following table describes the fields.
| Field Label | Description/Instructions |
|---|---|
Amendment Number | Enter an appropriate number. |
| Amendment Date* | Select or enter an appropriate date. |
You can select a different disease code only if accrual has not yet been reported to CTRP.
A trial can capture program codes from different organization families. For example, a participating site might belong to a different organization family than the lead organization. When you amend a trial, the Program Code field displays all codes from the master list for the organization family of the lead organization.
Primary Completion Dates are optional for non-interventional trials. The system excludes such trials when submitting XML documents to ClinicalTrials.gov. Otherwise, Primary Completion Dates are required.
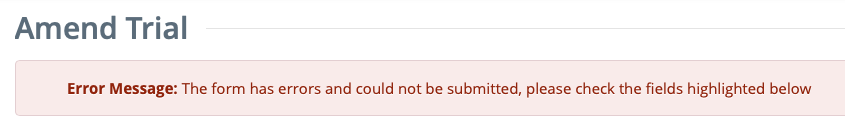
Correct any errors if indicated, and re-submit the amendment as many times as necessary until the amendment is error-free.
If no errors are found: The Trial Details page is loaded with a confirmation message at the top of the page: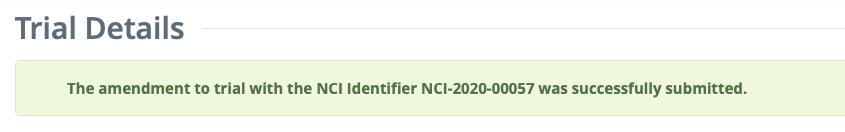
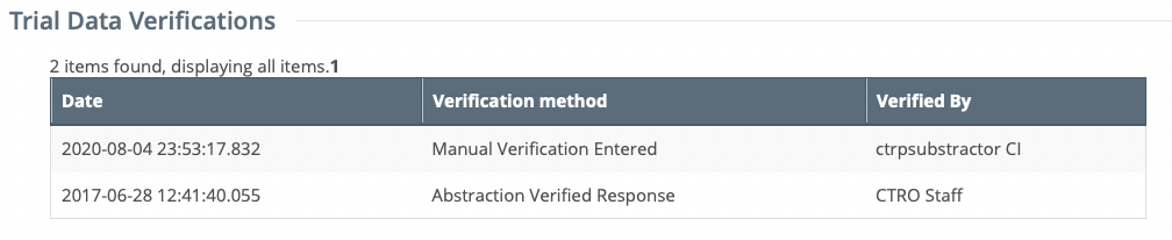
Each time a trial is amended, the CTRP Registration application records the Date, Verification Method and Verified By in the Trial Data Verifications history after the Amendments has been abstracted by the CTRO. The Trial Data Verifications history can be found on the Trial Details and Update/Verify Trial Data pages.