Search for the trial of interest. For instructions, refer to Searching for Trials in PA.
In the search results, select the NCI Trial Identifier link for the desired trial. This will open the Trial Identification page.
On the Trial Identification page, check out the trial. For instructions, refer to Checking In and Checking Out Trials. (This checkout step is optional for Super Abstractors.)
On the Administrative Data menu, select Participating Sites.
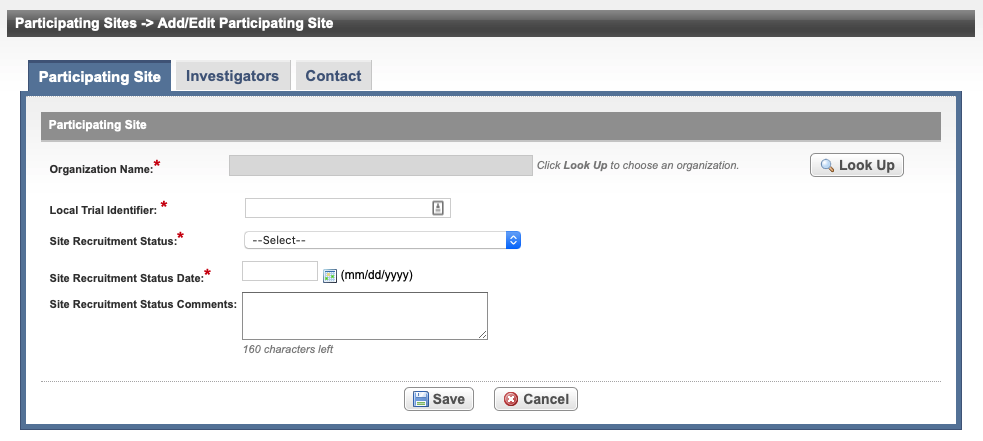
On the Participating Sites page, select Add.
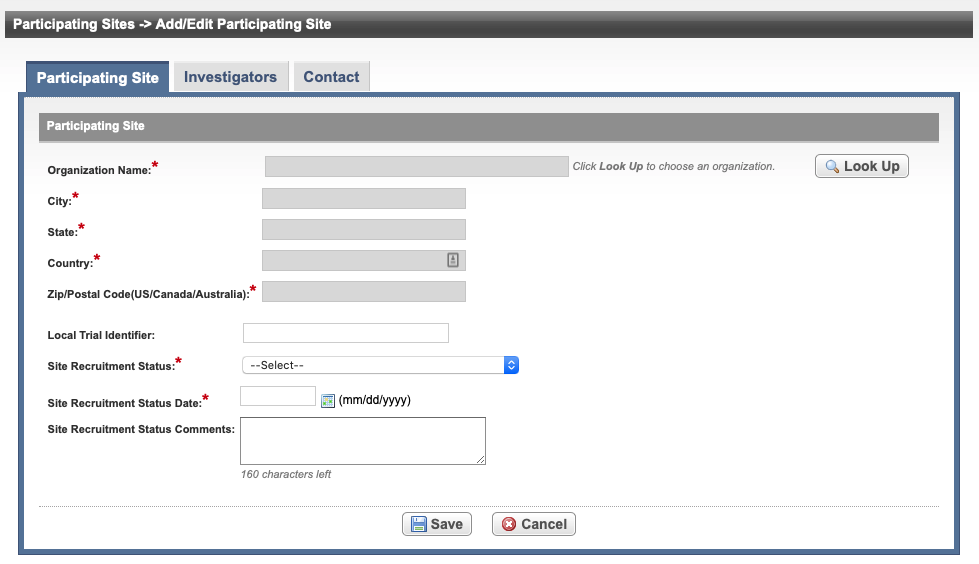
The Participating Sites page displays three tabbed sections: Participating Site, Investigators, and Contact.
|
See the following table for reference on the Participating Site fields. An asterisk (*) indicates a required field.
Field Label | Description/Instructions | |
|---|---|---|
Organization Name* | Select Look Up and follow the instructions in Searching for Organizations. For Complete trials, the City, State, Country, and Zip/Postal Codes fields are populated when an organization is selected. | |
Local Trial Identifier* | Enter the site trial identifier. | |
Site Recruitment Status* | Select the status from the drop-down list. Refer to Trial Status Values in the CTRP and ClinicalTrials.gov and Expanded Access Statuses for additional details regarding
| |
Site Recruitment Status Date* | Enter the date that the status was recorded. The date must be the current date or earlier. | |
| Site Recruitment Status Comments | Enter one or more comments about the site recruitment status. |
Select Save.
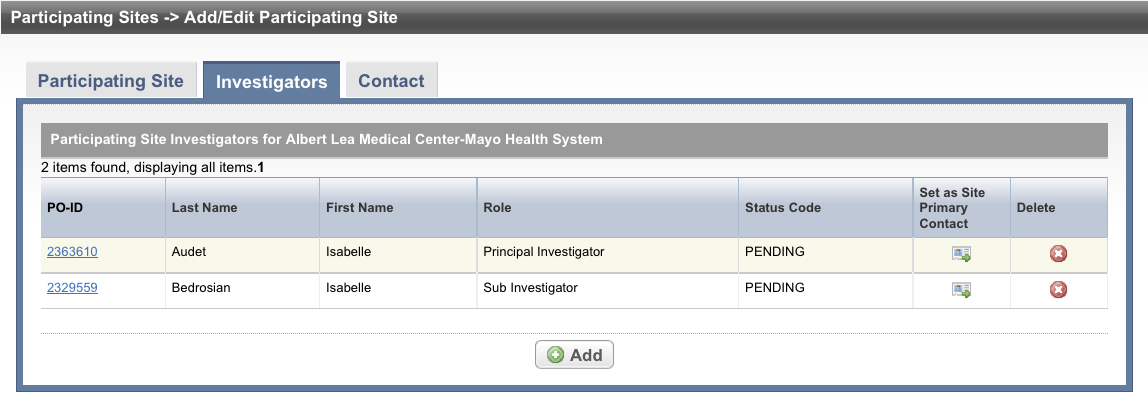
Select the Investigators tab. The Investigators tab displays the trial investigators that may have been added during trial submission or abstraction.
|
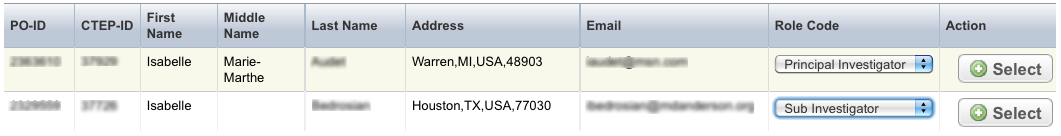
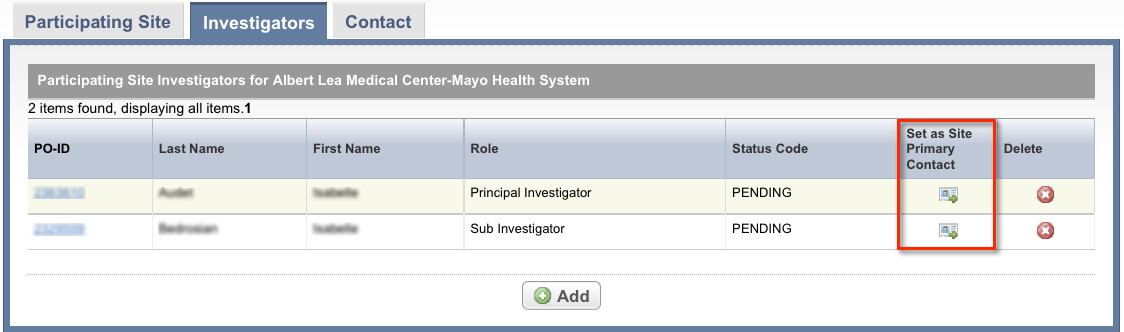
To indicate that an investigator is the primary contact, select the Set as Site Primary Contact icon next to this investigator's record.
Select the Contact tab.
A Participating Site must be linked to the trial before abstracting the site PI and contact information. A contact can be added by providing a person’s name (i.e., someone who is associated with the trial itself), or a generic contact can be added (i.e., someone who is associated with the site but not necessarily the trial) by providing a person’s title (functional role). Both types of contacts can not be added in the same record. An abstraction cannot be completed if a primary contact is not assigned. |
If an investigator is designated as the primary contact (on the Investigators tab), the investigator's name is displayed on the Contact tab. 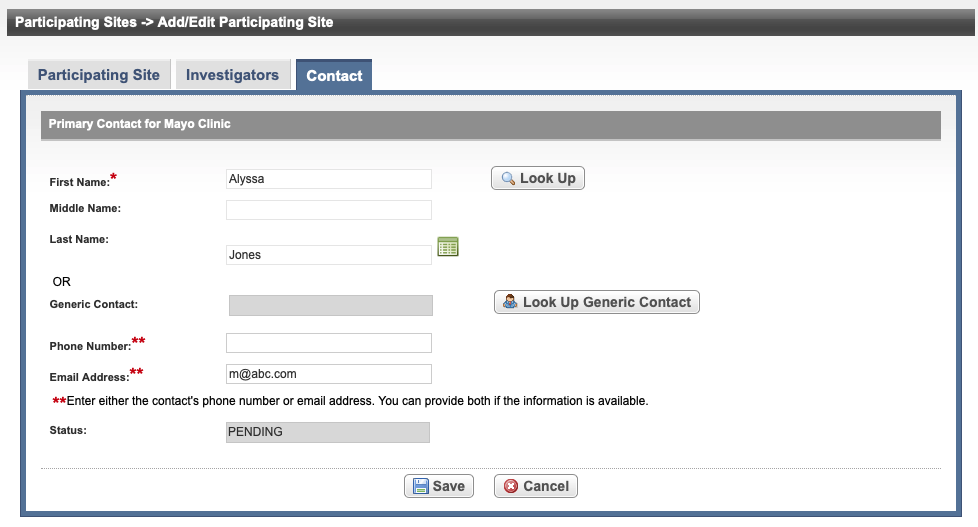
In the various fields, specify the appropriate information. The following table describes the fields. An asterisk (*) indicates a required field.
Field Label | Description/Instructions |
|---|---|
Phone Number* | Enter the contact’s primary telephone number (as 123-456-7890), including an extension if provided. *Either the contact's phone number or email address are required. Both can be provided. |
Email Address* | Type the contact’s primary email address. *Either the contact's phone number or email address are required. Both can be provided. |
| Status | CTRP populated after the record is saved. |