The Institutional Review Board (IRB) section captures IRB contact information which is optional, however this information is required to upload a study from CTRP to ClinicalTrials.gov.
How to Update Institutional Review Board (IRB) Information
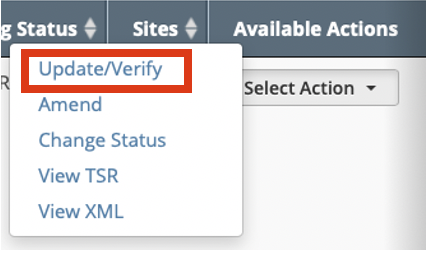
In the Available Actions column, click Select Action > Update/Verify. The Update Trial window lists the previously recorded IRB
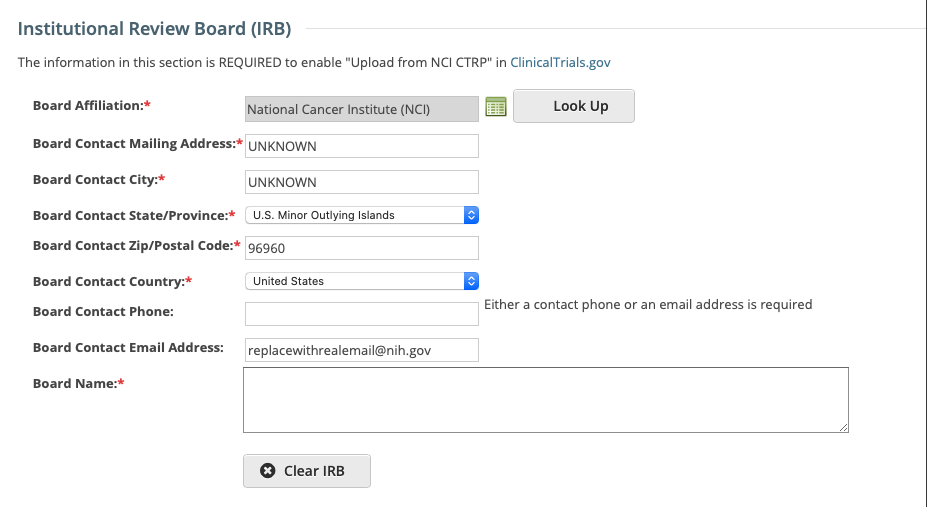
Select or enter the appropriate information in the text fields and drop-down lists. The following table describes the fields.
Field Label | Description/Instructions |
|---|---|
Board Affiliation* | Click Look Up to search for and select the Board name. If the name is not listed, register it with the CTRO. See Searching for Organizations. |
Board Name | The name of the affiliated organization. |