The Institutional Review Board (IRB) section captures IRB contact information which is optional, however this information is required to upload a study from CTRP to ClinicalTrials.gov.
How to Update Institutional Review Board (IRB) Information
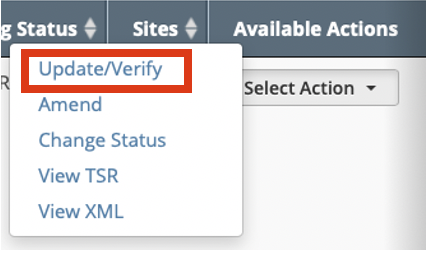
In the Available Actions column, click Select Action > Update/Verify. The Update Trial window lists the previously recorded IRB.
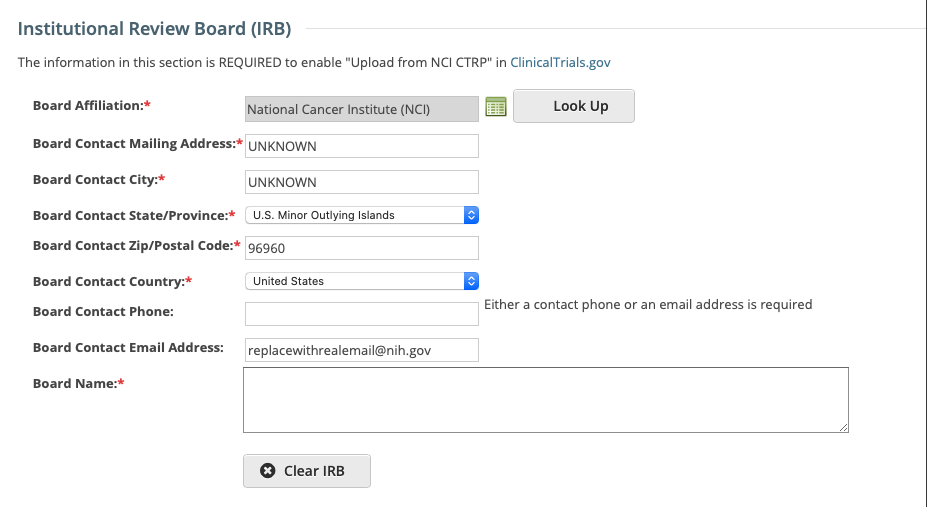
To select a new Board, use the Look Up button and follow the instructions on the Organization search page. The various Board Contact fields are pre-populated for the selected Board, but can be updated if desired, required fields are indicated with and asterisk (*).