Subject Disease Code (22)
For trials using ICD-O-3 terminology, use this position for Site and/or Histology Codes. When using both Site and Histology codes, separate them by a semicolon ( ; ). |
The following table lists the data fields by the table in which they are recorded. It also provides the field number (field position in a row); and the format and character limits for each field.
| Tables | Field (Field Position in a Row) | Format (Maximum Number of Characters) | |
|---|---|---|---|
| COLLECTIONS | Study Identifier (2) | Text (35) | |
| COLLECTIONS | Change Code (11) | Number (1) | |
| PATIENTS | Study Identifier (2) | Text (35) | |
| PATIENTS | Study Subject Identifier (3) | Text (20) | |
| PATIENTS | ZIP Code (4) | Text (10) | |
| PATIENTS | Country of Residence (5) | Text (2) | |
| PATIENTS | Patient's Date of Birth (6) | Date (YYYYMM) | |
| PATIENTS | Gender of a Person (7) | Text (10) | |
| PATIENTS | Ethnicity (8) | Text (25) | |
| PATIENTS | Payment Method (9) | Text (50) | |
| PATIENTS | Subject Registration Date (10) | Date (YYYYMMDD) | |
| PATIENTS | Registering Group Identifier (11) | Text (25) | |
| PATIENTS | Study Site Identifier (12) | Text (25) | |
| PATIENTS | Subject Disease Code (22)
| Number (10) | |
| PATIENT_RACES | Study Identifier (2) | Text (35) | |
| PATIENT_RACES | Study Subject Identifier (3) | Text (20) | |
| PATIENT_RACES | Race (4) | Text (45) |
The relation between COLLECTIONS, PATIENTS, and PATIENT_RACES is that COLLECTIONS can have multiple PATIENTS, and PATIENTS can have multiple PATIENT_RACES. These relations are maintained through the use of consistent Study Identifier and Study Subject Identifier fields. |
Using the format table, you can determine that you must enter the Patient's Date of Birth in the sixth position in a row in the PATIENTS table as YYYYMM. The following diagram contains a typical line of text from a batch file. Brackets with numbers indicate the field position number.
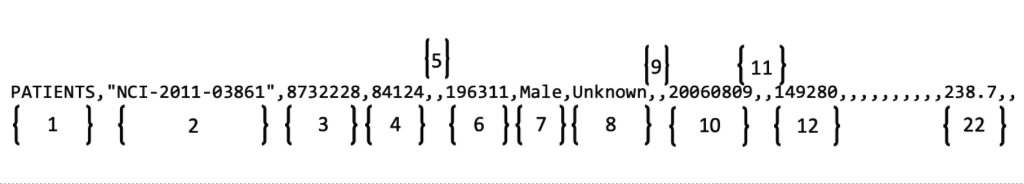
In the example above,
|
___
For trials using ICD-O-3 terminology, use this position for Site and/or Histology Codes. When using both Site and Histology codes, separate them by a semicolon ( ; ).