The Trial Eligibility Criteria information provides a summary of the qualifications for subject enrollment in the study. It is optional for Abbreviated trials. If you choose to record trial eligibility criteria for Abbreviated trials, be sure to complete all fields.
Eligibility criteria include a set of demographic information (i.e., health, gender, age) and other eligibility criteria in a table consisting of both “Inclusion” and “Exclusion” criteria.
Abstracting Interventional Trial Eligibility Criteria
Search for the trial of interest. For instructions, refer to Searching for Trials in PA.
In the search results, click the NCI Trial Identifier link for that trial. The Trial Identification page appears.
On the Trial Identification page, check out the trial. For instructions, refer to Checking In and Checking Out Trials. (This checkout step is optional for Super Abstractors.)
On the Scientific Data menu, under Interventional Trial Design, click Eligibility Criteria. The Interventional Trial Design – Eligibility Criteria page appears.
If the trial has been abstracted previously, the Eligibility Criteria section displays the demographic eligibility elements. The Other Criteria section may also display inclusion and/or exclusion criteria that have been added previously (see Adding Other Eligibility Criteria).- In the drop-down lists and text fields, select or enter the appropriate information. The following table describes the fields. All fields are required.
Field
Instruction
Accepts Healthy Volunteers*
If the trial accepts healthy participants, select Yes. Otherwise, select No.
Sex*
Indicate the sex of the individuals who may participate in the clinical trial:
- Male: Indicates that the trial studies only male participants.
- Female: Indicates that the trial studies only female participants.
- All: Indicates that the recruitment criteria do not limit eligibility based on the sex of participants.
If you select Female or Male in this field, the system allows you to specify a value in the Gender Based field.
Gender Based
If participant eligibility is based on self-representation of gender identity, select Yes. Otherwise, select No. If you select Yes in this field, the optional Gender Eligibility Description field becomes available. Gender Eligibility Description Enter any available information about gender eligibility. Minimum Age*
Enter the minimum participant age. If there is no minimum age, type 0 , and select Years from the Unit drop-down list.
Maximum Age*
Enter the maximum participant age. If there is no maximum age, type 999 , and select Years from the Unit drop-down list.
Unit*
Select the age unit from the drop-down list. Valid values are as follows:
- Years
- Months
- Weeks
- Days
- Hours
- Minutes
You can expand the Other Criterion table columns to read full criteria descriptions and names
To increase the size of a description or name cell, click and drag the lower right corner handles.
Original cell size with hidden text
Expanded cell revealing all text - Click Save.
The trial is updated with the information you provided, and a new interface appears which enables you to add other criteria. See Adding Other Eligibility Criteria.
Editing Demographic Eligibility Criteria
Search for the trial of interest. For instructions, refer to Searching for Trials in PA.
In the search results, click the NCI Trial Identifier link for that trial. The Trial Identification page appears.
On the Trial Identification page, check out the trial. For instructions, refer to Checking In and Checking Out Trials. (This checkout step is optional for Super Abstractors.)
On the Scientific Data menu, under Interventional Trial Design or Non-interventional Trial Design, click Eligibility Criteria. The Eligibility Criteria page appears.
- Modify the information as necessary, and then click Save.
Abstracting Non-Interventional Trial Eligibility Criteria
Search for the trial of interest. For instructions, refer to Searching for Trials in PA.
In the search results, click the NCI Trial Identifier link for that trial. The Trial Identification page appears.
On the Trial Identification page, check out the trial. For instructions, refer to Checking In and Checking Out Trials. (This checkout step is optional for Super Abstractors.)
On the Scientific Data menu, under Non-interventional Trial Design, click Eligibility Criteria. The Non-Interventional Trial Design – Eligibility Criteria page appears.
If the trial has been abstracted previously, the Eligibility Criteria section displays the demographic eligibility elements. The Other Criteria section may also display inclusion and/or exclusion criteria that have been added previously (see Adding Other Eligibility Criteria).- In the drop-down lists and text fields, select or enter the appropriate information. The following table describes the fields. All fields are required.
Field
Instruction
Accepts Healthy Volunteers*
If the trial accepts healthy participants, select Yes. Otherwise, select No.
Sex*
Indicate the sex of the individuals who may participate in the clinical trial:
- Male: Indicates that the trial studies only male participants.
- Female: Indicates that the trial studies only female participants.
- All: Indicates that the recruitment criteria do not limit eligibility based on the sex of participants.
If you select Female or Male in this field, the system allows you to specify a value in the Gender Based field.
Gender Based
If participant eligibility is based on self-representation of gender identity, select Yes. Otherwise, select No. If you select Yes in this field, the optional Gender Eligibility Description field becomes available. Gender Eligibility Description Enter any available information about gender eligibility. Minimum Age*
Enter the minimum participant age. If there is no minimum age, type 0 , and select Years from the Unit drop-down list.
Maximum Age*
Enter the maximum participant age. If there is no maximum age, type 999 , and select Years from the Unit drop-down list.
Unit*
Select the age unit from the drop-down list. Valid values are as follows:
- Years
- Months
- Weeks
- Days
- Hours
- Minutes
In the additional fields for non-interventional trials, select or enter the appropriate information. The following table describes the fields. All fields are required.
Field
Instruction/Description
Sampling Method* Select the sampling method from the drop-down list. Valid values are as follows:
- Probability Sample. Exclusively random process to guarantee that each participant or population has specified chance of selection, such as simple random sampling, systematic sampling, stratified random sampling, cluster sampling, and consecutive patient sampling
- Non-Probability Sample. Any of a variety of other sampling processes, such as convenience sampling or invitation to volunteer
Study Population Description* Describe the population from which the groups or cohorts will be selected (e.g., primary care clinic, community sample, residents of a certain town).
You can expand the Other Criterion table columns to read full criteria descriptions and names
To increase the size of a description or name cell, click and drag the lower right corner handles.
Original cell size with hidden text
Expanded cell revealing all text- Click Save.
The trial is updated with the information you provided, and a new interface appears which enables you to add other criteria.
Adding Other Eligibility Criteria
How to Add Eligibility Criteria
Search for the trial of interest. For instructions, refer to Searching for Trials in PA.
In the search results, click the NCI Trial Identifier link for that trial. The Trial Identification page appears.
On the Trial Identification page, check out the trial. For instructions, refer to Checking In and Checking Out Trials. (This checkout step is optional for Super Abstractors.)
On the Scientific Data menu, under Interventional Trial Design or Non-interventional Trial Design, click Eligibility Criteria. The Eligibility Criteria page appears. If the Add Another Criterion button is not displayed, complete all demographic fields on the Eligibility page first.
- Click Add Another Criterion.
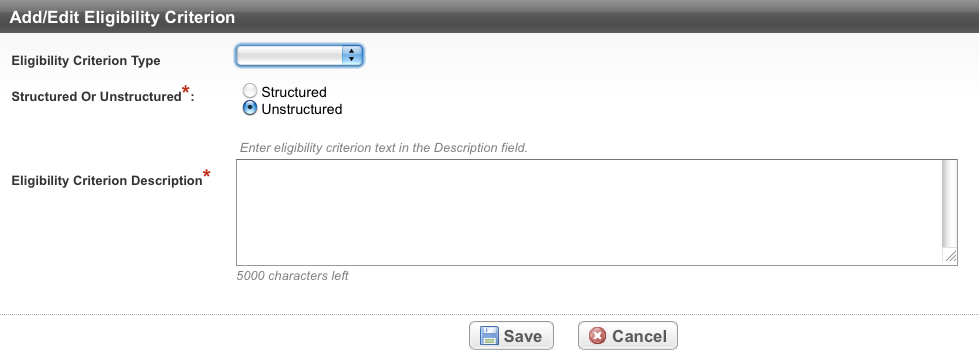
The Add/Edit Eligibility Criterion Page appears.
In the drop-down lists and text fields, select or enter the appropriate information. The following table describes the fields. An asterisk (*) indicates a required field.
Field
Instruction/Description
Eligibility Criterion Type
Indicate whether satisfying the criterion qualifies or disqualifies a subject from participation in the trial by selecting Inclusion or Exclusion respectively.
Structured or Unstructured*
The Unstructured option is preselected. This is the only method by which you can record eligibility criteria via Protocol Abstraction.
Eligibility Criterion Description*
Enter a comprehensive description of the criterion.
- Click Save.
The new criterion record is displayed on the Eligibility Criteria page.
Editing Other Trial Eligibility Criteria
Search for the trial of interest. For instructions, refer to Searching for Trials in PA.
In the search results, click the NCI Trial Identifier link for that trial. The Trial Identification page appears.
On the Trial Identification page, check out the trial. For instructions, refer to Checking In and Checking Out Trials. (This checkout step is optional for Super Abstractors.)
On the Scientific Data menu, under Interventional Trial Design or Non-interventional Trial Design, click Eligibility Criteria. The Eligibility Criteria page appears.
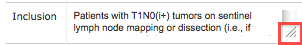
- In the Edit column for the record you want to modify, click the Edit (pencil) icon.
The Add/Edit Eligibility Criterion page appears, displaying the criterion as submitted. - Modify any or all of the information, and then click Save.
Reordering Other Eligibility Criteria
How to Re-Order Other Eligibility Criteria
Search for the trial of interest. For instructions, refer to Searching for Trials in PA.
In the search results, click the NCI Trial Identifier link for that trial. The Trial Identification page appears.
On the Trial Identification page, check out the trial. For instructions, refer to Checking In and Checking Out Trials. (This checkout step is optional for Super Abstractors.)
On the Scientific Data menu, under Interventional Trial Design or Non-interventional Trial Design, click Eligibility Criteria. The Eligibility Criteria page appears, displaying all other eligibility criteria currently abstracted.
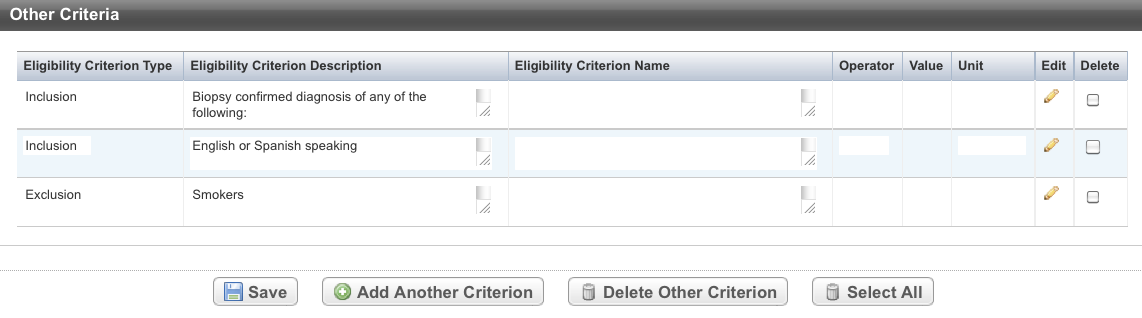
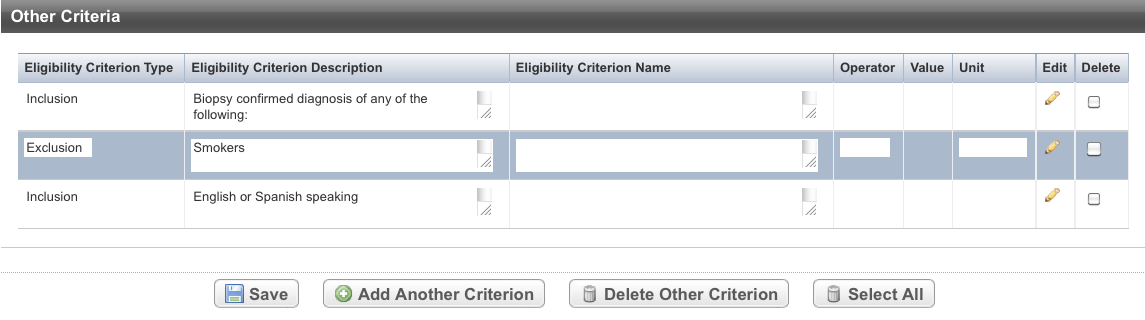
- Hover your mouse in the Eligibility Criterion Type column.
The cursor changes shape. - Click and drag the record you want to move to a new location in the Other Criteria table. The record background changes to grey as you move it.
The system displays a message to confirm that the record has been updated.
Deleting Eligibility Criteria
How to Delete Eligibility Criteria
Search for the trial of interest. For instructions, refer to Searching for Trials in PA.
In the search results, click the NCI Trial Identifier link for that trial. The Trial Identification page appears.
On the Trial Identification page, check out the trial. For instructions, refer to Checking In and Checking Out Trials. (This checkout step is optional for Super Abstractors.)
On the Scientific Data menu, under Interventional Trial Design or Non-interventional Trial Design, click Eligibility Criteria. The Eligibility Criteria page appears.
In the Delete column for the record you want to delete, click the Delete icon.
Warning
There is no way to recover a deleted record. If you delete a record accidentally, follow the instructions in Adding Other Eligibility Criteria to add it back.