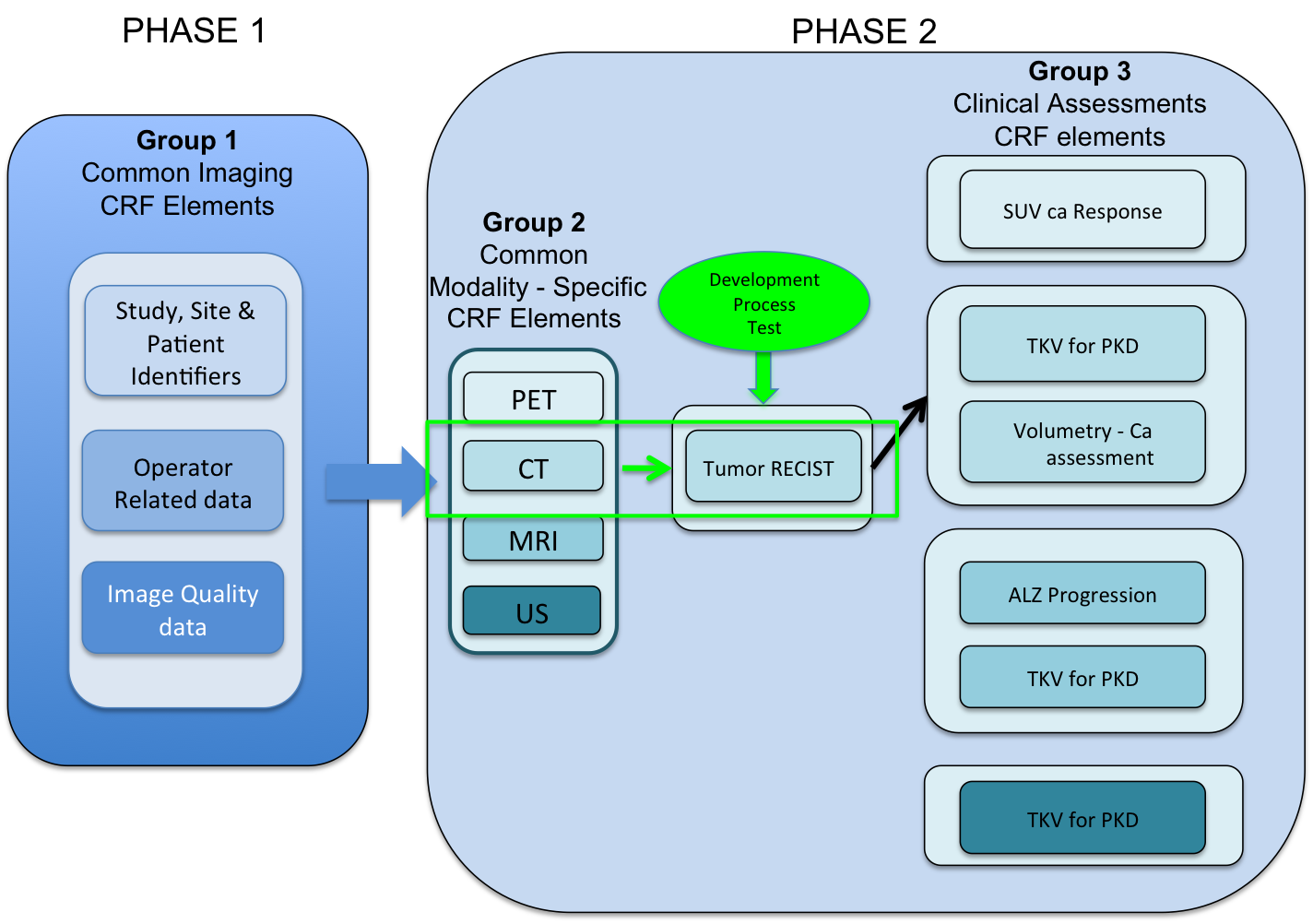
Project Scope and Goals
A collaborative project between NCI (EVS and caDSR) and CDISC with the goal of identifying a core set of imaging variables that can be combined with clinical elements to produce a harmonized set that supports clinical care, regulatory submission(s) and electronic health records. The set will support established imaging and clinical data standards. The focus of this project is to point to and map across existing standards.
These established imaging and clinical data standards include: BRIDG, AIM, ACRIN, NBIA, CIP, QIN, QIBA, DICOM, caDSR, ISO data types, RadLex, NCIt, CDISC Controlled terminology, CDASH, SDTM, Hl7 and meaningful use standards.
The first use case comes from the Imaging workspace and is focused on the CT modality and RECIST 1.1.
Imaging Project Plan Charter Document 2011 06 02
Project Plan
Mapping Spreadsheet
Imaging Metadata CT RECIST Spreadsheet 2011 08 09 jc
Imaging Metadata CT RECIST Spreadsheet 2011_12-06
Meeting Notes
Team Members
•Dianne Reeves (NCI) •Rhonda Facile (NCI-CDISC) •Janice Chillin (SIAC) •Tina Taylor (ACRIN) •Andy Buckler (BBMSC) •Jesse Bowden (BioMedical Systems) •Liwei Wu (BioMedical Systems) •Jessica Nassker (Deloitte) •LeaAnn Bailey (Deloitte) •Jon Neville (C-Path) •Chris Tolk (CDISC) •Ed Helton (NCI) •Elliot Segal (U of Maryland) •Enrique Aviles (C-Path) •Frank Newby (CDISC) •Shannon Labout (CDISC)
TC Schedule and Dial-In Information
Every 2 weeks on Thursdays at 3pm EDT starting January 12, 2012
ADOBE Connect: http://cdisc.adobeconnect.com/imagingcrf/
Dial in: 1 415 363 0833 PIN: 563707