Introduction
The CRF Harmonization and Standardization Initiative was launched in 2006 based on recommendations from the Clinical Trials Working Group (CTWG) report of 2005. The CTWG recommended developing standardized Case Report Forms incorporating Common Data Elements to improve information sharing among cancer researchers and optimize data requirements in collaboration with the Federal Drug Administration (FDA). A harmonized CRF library will improve the efficiency and accuracy of the routine review of safety, efficacy, and administrative data from ongoing NCI-funded clinical trials. Finally, by reducing the time spent in developing a data collection strategy per trial, this core library will allow for faster initiation of new trials; thus, speeding the process of delivering new and improved treatments to patients.
The Standardized CRF Work Group was originally part of the Study Conduct Special Interest Group (SIG) and the Clinical Trials Management Systems (CTMS) Workspace.
Prioritizing the work
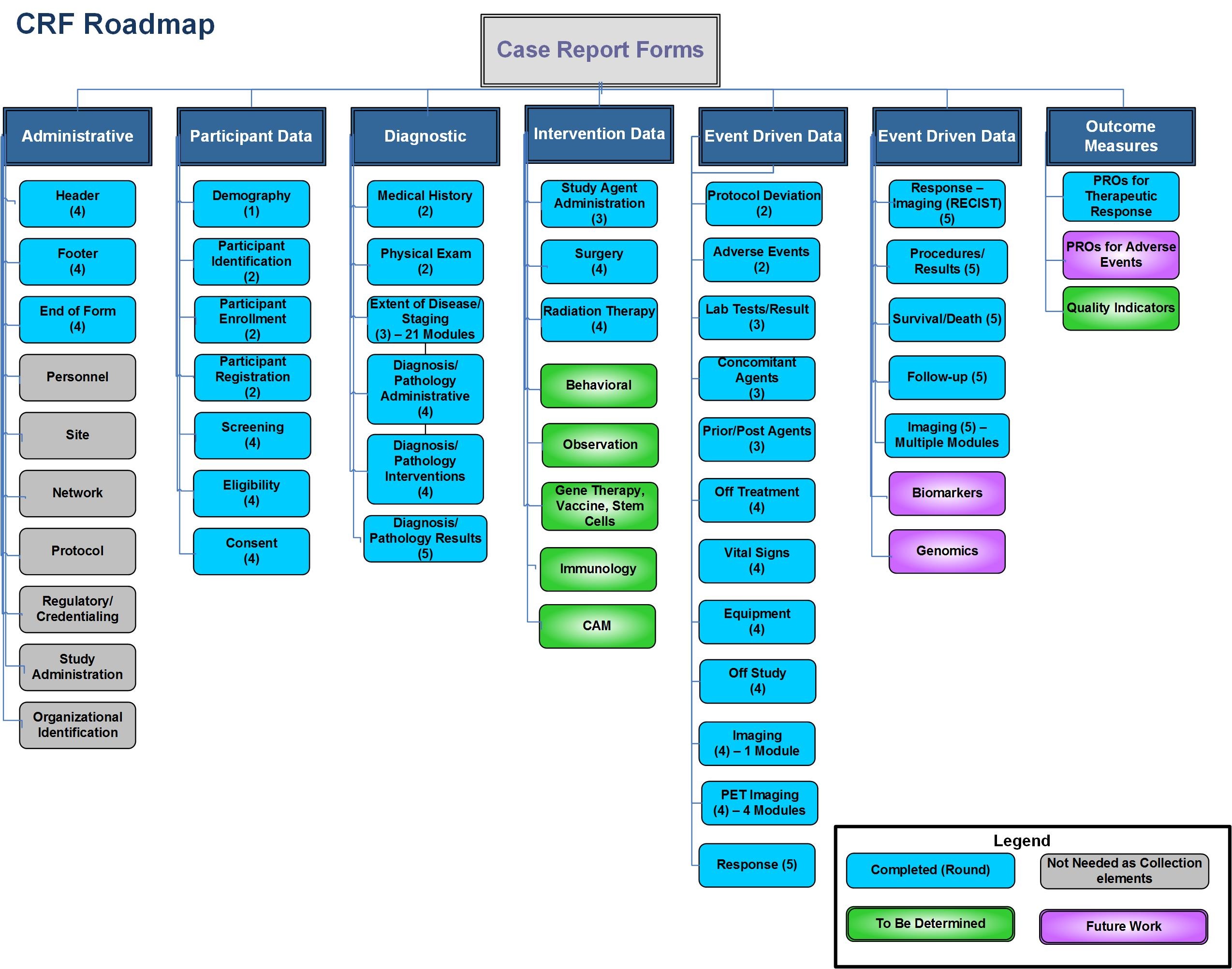
To begin the CRF work, a road map was created that has evolved over time to reflect the progress made by the community. The prioritization started by choosing one module that could be a proof of concept or a test of process for a way the work group could be structured. The first module chosen was the Demography module. As the team worked through this module defining structures, group rules, interactions, and best practices they begin to move forward on areas of content such as Adverse Events and Enrollment. The leads for each of the working groups remained the same for consistency. The road map continues to evolve and will be completed when a set of elements have been identified to reflect the core data collection and analysis points for a clinical trial.
- Brenda Duggan, Manager, Community Informatics Program, NCI CBIIT
- Neesha Desai, CRF Project Manager, NCI CBIIT (Contractor)