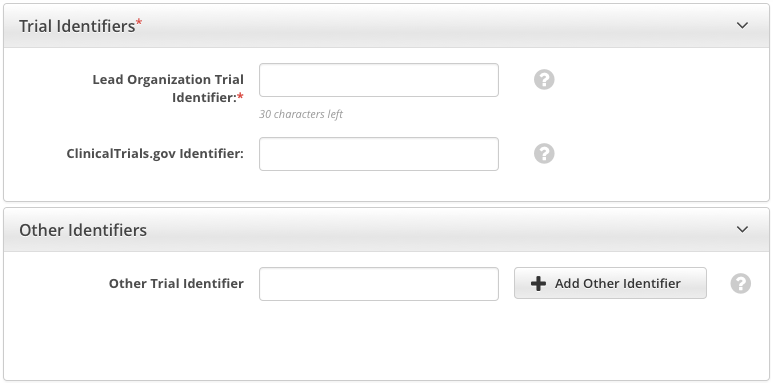
How to Complete the Trial Identifiers Section
Select or enter the appropriate information in the text fields and drop-down lists. The following table describes the fields. An asterisk (*) indicates a required field.
Instructions for recording Trial Identifiers for Complete trials
Field Label | Description/Instructions |
|---|---|
Lead Organization Trial Identifier* | Enter the unique identification assigned to the protocol by the sponsoring organization, exactly as it appears in the protocol document. For Inter-Group trials, type the Lead Group's trial number. For multi-site trials that have no assigned single center, use the protocol ID, for example, |
ClinicalTrials.gov Identifier | If the trial has been submitted to ClinicalTrials.gov previously, enter the number assigned to the trial by PRS (ClinicalTrials.gov). The CTRP renamed the previous identifiers "NCT Number" and "NCT ID" to "ClinicalTrials.gov Identifier". You cannot change the ClinicalTrials.gov Identifier once you have added it. If you need to make changes thereafter, contact the CTRO at NCICTRO@mail.nih.gov. When you submit the trial, the system checks the ClinicalTrials.gov identifier you entered to ensure that no other registered trial has the same one. The system displays an error message if it finds another trial with the same ClinicalTrials.gov identifier. If this occurs, check the number you entered and try again. If you are certain that the number you entered is correct, contact the CTRO at NCICTRO@mail.nih.gov. |
Other Trial Identifier | Enter an additional trial identifier such as unique identifier from other registries, NIH grant numbers, or protocol numbers assigned by the Review Board, and then click Add Other Identifier. Repeat this step for each additional identifier. |