Valid trial statuses used in CTRP are mapped to ClinicalTrials.gov-defined recruitment values. For valid ClinicalTrials.gov recruitment values, refer to Trial Status Values in the CTRP and ClinicalTrials.gov and Expanded Access Statuses.
If you register a trial after it has reached any Closed to Accrual related trial status or a Complete trial status, you must create a complete Trial Status History by adding all previous statuses in order, as per the rules provided in Status Transition Rules.
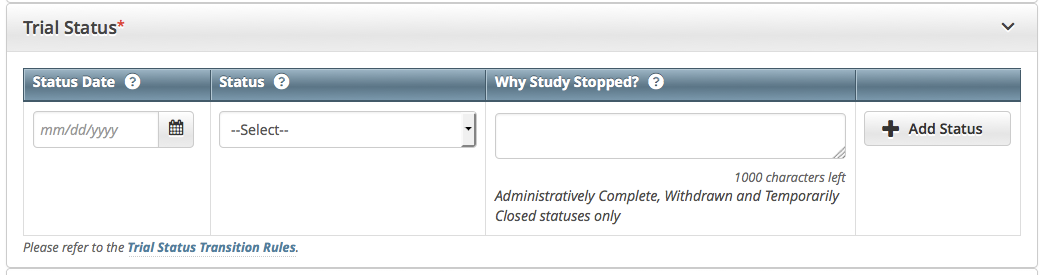
How to Complete the Trial Status Section
Select or enter the appropriate information in the text fields and drop-down lists. The following table describes the fields. All fields in this section are required.
Field Label
Description/Instructions
Trial Status Date*
Enter the date on which the current trial status became effective.
Trial Status
Select the trial’s accrual status from the drop-down list. For valid values, refer to Trial Status Values in the CTRP and ClinicalTrials.gov and Expanded Access Statuses.
Why Study Stopped
If you selected the Administratively Complete, Withdrawn, or Temporarily Closed to Accrual status, type the reason why the study has ended or is not currently accruing, using up to 160 characters. As you type, the number of characters remaining is displayed below the text field.
Click Add Status.
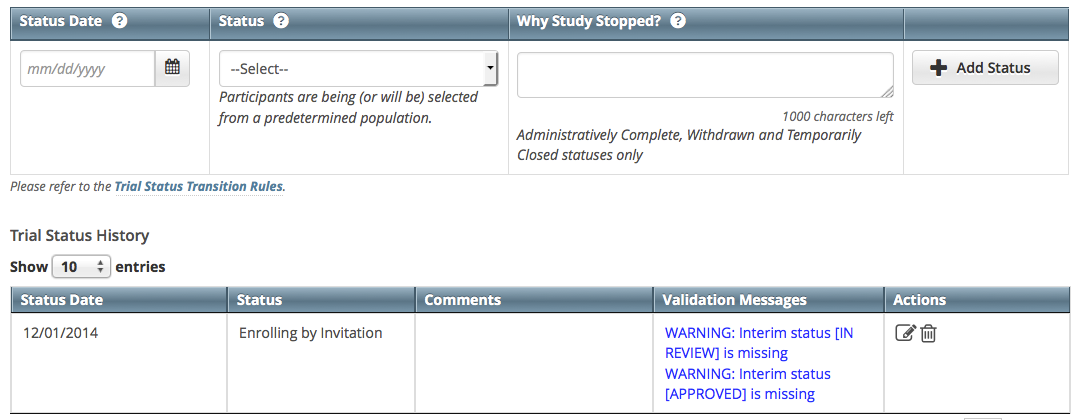
The new status record is displayed.- If the system indicates that the status you added is invalid, do one of the following.
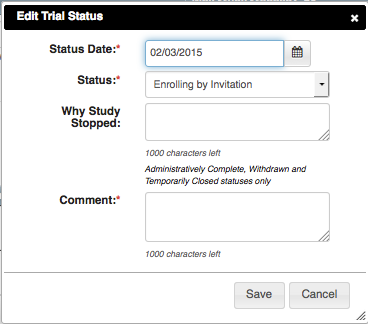
- To edit the status, in the Actions column, click the Edit icon and make changes as indicated in the Error and/or Warning message.
- To delete the status, in the Actions column, click the Delete icon. Enter a comment indicating the reason why you deleted the record, and then add the correct status information.