- Submission Received Date
- Submission Acceptance Date
- Submission Reactivated Date
- Administrative Processing Start Date
- Administrative Processing Completed Date
- Ready for Administrative QC Date
- Administrative QC Start Date
- Administrative QC Completed Date
- Scientific Processing Start Date
- Scientific Processing Completed Date
- Ready for Scientific QC Date
- Scientific QC Start Date
- Scientific QC Completed Date
- Ready for Trial Summary Report Date
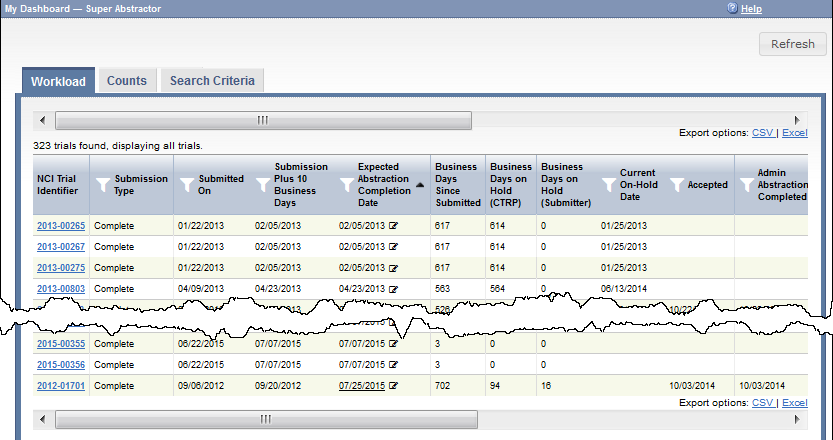
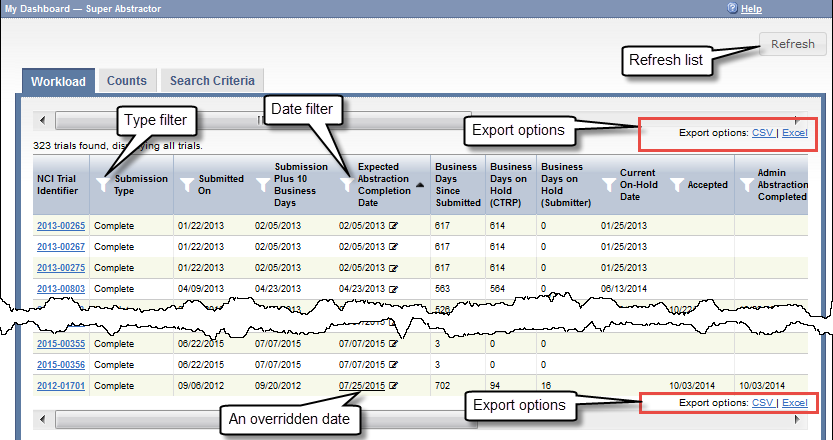
The Workload page provides up-to-the-minute details of each trial. The following table describes each column on the Workload page:
| Column in Workload Page | Description |
|---|---|
| NCI Trial Identifier | The unique ID assigned to the trial by the CTRP. NCI Trial Identifiers The prefix "NCI" is implicit in the NCI trial identifiers |
| Submission Type | The submission type:
For information, refer to Processing Trial History Information (original versus amendment) or Abstracting NCI-Specific Information (Complete versus Abbreviated). |
| Submitted On | The date on which the CTRP user submitted the trial. |
| Submission Plus 10 Business Days | A calculated date, available only to Super Abstractors. This calculation starts with the Submitted date, adds ten business days, and adds any number of days in which trial was on hold waiting for information from the submitter. The system requires this date to fall on a business day. This date is a good starting point in deciding how soon abstraction should be completed for each trial. (A business day is any weekday that is not a Federal holiday. For a list of Federal holidays, refer to the U.S. Office of Personnel Management's list of Federal Holidays.) |
| Expected Abstraction Completion Date | A calculated date, by default. The system calculates this date as follows:
(A business day is any weekday that is not a Federal holiday. For a list of Federal holidays, refer to the U.S. Office of Personnel Management's list of Federal Holidays.) If the date has an underline, a Super Abstractor has overridden the default date for the trial. |
| Business Days Since Submitted | A calculated value, available only to Super Abstractors. This calculation starts with today, subtracts the Submitted date, and subtracts any weekend days and Federal holidays between (and including) the two dates. |
| Business Days on Hold (CTRP) | A calculated value, available only to Super Abstractors. If a value appears in this column for a trial, the system calculated it based on the Reason and Reason Category selected each time an abstractor put the trial on hold: This calculation is the total number of days in which the trial has been on hold, waiting for information from CTRP. |
| Business Days on Hold (Submitter) | A calculated value, available only to Super Abstractors. If a value appears in this column for a trial, the system calculated it based on the Reason and Reason Category selected each time an abstractor put the trial on hold: This calculation is the total number of days in which the trial has been on hold, waiting for information from the submitter. |
| Current On-Hold Date | The on-hold date, if the trial is on-hold today. For details, refer to Putting Trial Processing On Hold. |
| Accepted | The date of the Submission Acceptance Date milestone, if the trial has reached that point. For details, refer to Processing Trial Milestones. |
| Admin Abstraction Completed | The date of the Administrative Processing Completed Date milestone, if the trial has reached that point. For details, refer to Processing Trial Milestones. |
| Admin QC Completed | The date of the Administrative QC Completed Date milestone, if the trial has reached that point. For details, refer to Processing Trial Milestones. |
| Scientific Abstraction Completed | The date of the Scientific Processing Completed Date milestone, if the trial has reached that point. For details, refer to Processing Trial Milestones. |
| Scientific QC Completed | The date of the Scientific QC Completed Date milestone, if the trial has reached that point. For details, refer to Processing Trial Milestones. |
| Ready for TSR | The date of the Ready for Trial Summary Report Date milestone, if the trial has reached that point. For details, refer to Processing Trial Milestones. |
| Checked Out By | The name of the user who has the trial checked out, if any. The Workload page also displays an abbreviation in parentheses indicating the type of check-out each user has for the trial:
For details, refer to Checking In and Checking Out Trials. |
How to Use the Workload Page
To do this... | Do this |
|---|---|
Sort the list of trials | By default, the system sorts the trials by Expected Abstraction Completion Date. To sort the list by any other column, click any column header. To reverse the order, click the column header again. An arrow indicates the sort order:
|
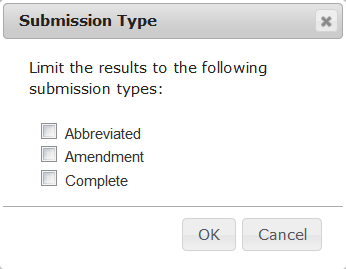
| Filter the list of trials by submission type | Click the Filter icon () in the Submission Type column. The Submission Type dialog box appears. Select one or more submission types that you want to include and click OK. |
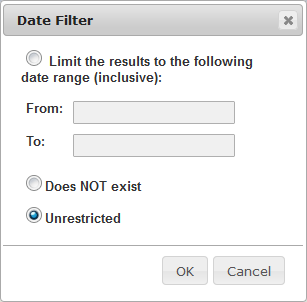
| Filter the list of trials by date |
|
Update the list of trials and reset all filters | Click the Refresh button. |
Export the list of trials to a file | On the upper or lower right corner of the page, click one of the following:
|
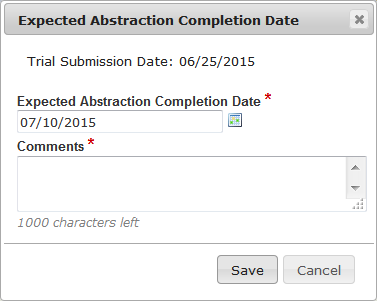
| Change the Expected Abstraction Completion Date for a trial |
|
| View comments on an overridden Expected Abstraction Completion Date | Hover the mouse over the underlined date. |
View trial details | Click an NCI Trial Identifier link. The Details page appears with information about the trial you selected. |