The ISO/IEC 11179 Information Technology: Metadata Registries (MDR) specification developed by the International Organization for Standardization and the IEC (International Electrotechnical Commission) defines a number of fields and relationships for Metadata Registries.
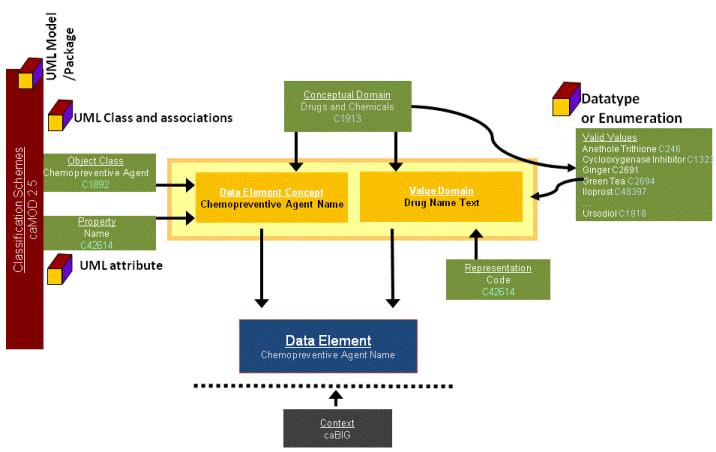
Included is a detailed metamodel for defining and registering administered items. The primary component of that metamodel is a Data Element. The diagram below illustrates how the various components of the metamodel relate to each other. Each box in the diagram contains the name of an ISO/IEC 11179 administered item type, and underneath an example of metadata describing the various parts of a CDE.
In a typical data dictionary a data element might consist of a name, datatype, length, and perhaps a definition. Sometimes an enumerated list of values is specified to constrain data entry. The preceding diagram shows that the data element embodies a much more extensive collection of carefully designed components and relationships that decompose and describe the essence of a data element in well formed parts, separating the conceptual entity (Data Element Concept) from its physical representation in a database (Value Domain). The "EVS" icons indicate where the NCI to date has linked controlled vocabularies to administered items in naming and defining these metadata, providing the foundation for computable, unambiguous semantic interoperability.
A Data Element is comprised of a Data Element Concept (the concept or meaning), and a Value Domain (the specific representation). Value Domains have a list of Valid (or Permissible) Values. Data Element Concepts may be associated with an Object Class and a Property; Value Domains form Representations. Each can be additionally modified with one or more Qualifier concepts. The terminology for naming and defining Object Class, Property, Representation, Qualfiers and Value Domains and Value Meanings comes from controlled vocabularies found in the Enterprise Vocabulary Services component of caCORE. Data Element Concepts and Value Domains each belong to a higher level Conceptual Domain.
The Context field is used to logically separate the various CDE development efforts going on at the NCI. The Cancer Therapy Evaluation Program (CTEP) Context contains the largest number of CDEs approved for use in NCI clinical trials.
Classification Schemes contain Classification Scheme Items that a Data Element can be assigned to. A Data Element can be assigned to any number of Classification Scheme Items, and Classification Scheme Items may be related other Classification Scheme Items, such as in a hierarchy. Only Classification Schemes with a "Released" workflow status are displayed in OneData.