|
Page History
...
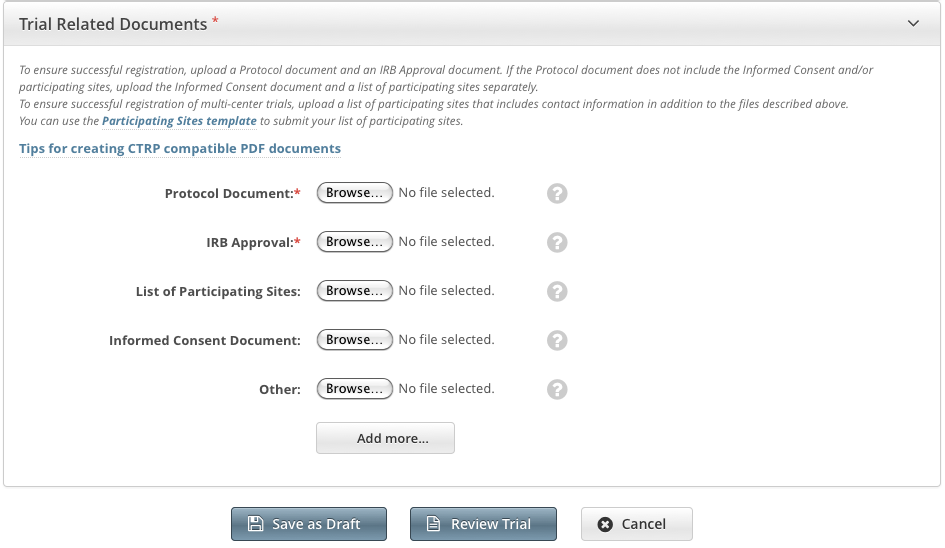
- Complete clean Protocol document.
- IRB Approval.
- List of Participating Sites (if not included in the protocol document). Multi-site trials require a list of participating sites and contact information.
Informed Consent (if not included in the protocol document).
Currently, the system requires you are required to supply each of your documents as one of the following formats:
- Microsoft Word (.doc, .docx, or .docm),.
- Adobe PDF. Adobe PDF files require special processing. For information about creating PDFs, refer to Converting Files to PDFs.
- Microsoft Excel (.xls, .xlsx, .xlsm, or .xlsb),.
- and/or WordPerfect files.
...
...
Adobe PDF files require special processing. For information about creating PDFs, refer to Converting Trial-Related Documents to PDFs.
How to Submit Trial Related Documents
- Next to the document-type field, (e.g., such as Protocol Document), click Browse.
Navigate Follow your browser/operating system instructions to navigate to, and select, and open the appropriate document, and then click Open.
infoDepending on your operating system, you may see a different command name for "Open."
Repeat the steps above for each type of document.
Tip title Adding Multiple "Other" Documents You can upload more than one (1) "Other" document. After you have uploaded the first of your "Other" documents, click the Add More link. A The system displays a new Other document field is displayed.