|
Page History
...
| Column | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| |||||||||
|
...
Module Status Table
...
Round of Harmonization & Standardization
...
CRF Modules
...
Current Status of Module
...
1
...
...
Harmonization complete; Standards presentation 9/3/09; Approved CDEs as standard on 10/30/09
...
2
...
...
Harmonization complete; preparing for standards review
...
2
...
...
Harmonization complete; preparing for standards review
...
2
...
...
Harmonization complete; preparing for standards review
...
2
...
...
Harmonization complete; preparing for standards review
...
2
...
...
Harmonization complete; preparing for standards review
...
2
...
...
Harmonization complete; preparing for standards review
...
2
...
...
Harmonization complete; preparing for standards review
...
3
...
...
Harmonization nearing completion
...
3
...
...
Harmonization nearing completion
...
3
...
...
Harmonization nearing completion
...
3
...
...
Harmonization nearing completion
...
3
...
...
Harmonization nearing completion
...
3
...
...
Harmonization nearing completion
...
3
...
...
Harmonization nearing completion
...
3
...
...
Harmonization nearing completion
...
3
...
...
Harmonization nearing completion
...
4
...
...
Harmonization is complete
...
4
...
...
Harmonization is complete
...
4
...
...
Harmonization is complete
...
4
...
...
Harmonization is complete
...
4
...
Non- Agent Study Interventions
...
Harmonization is complete
...
4
...
...
Harmonization is complete
...
4
...
...
Introduction
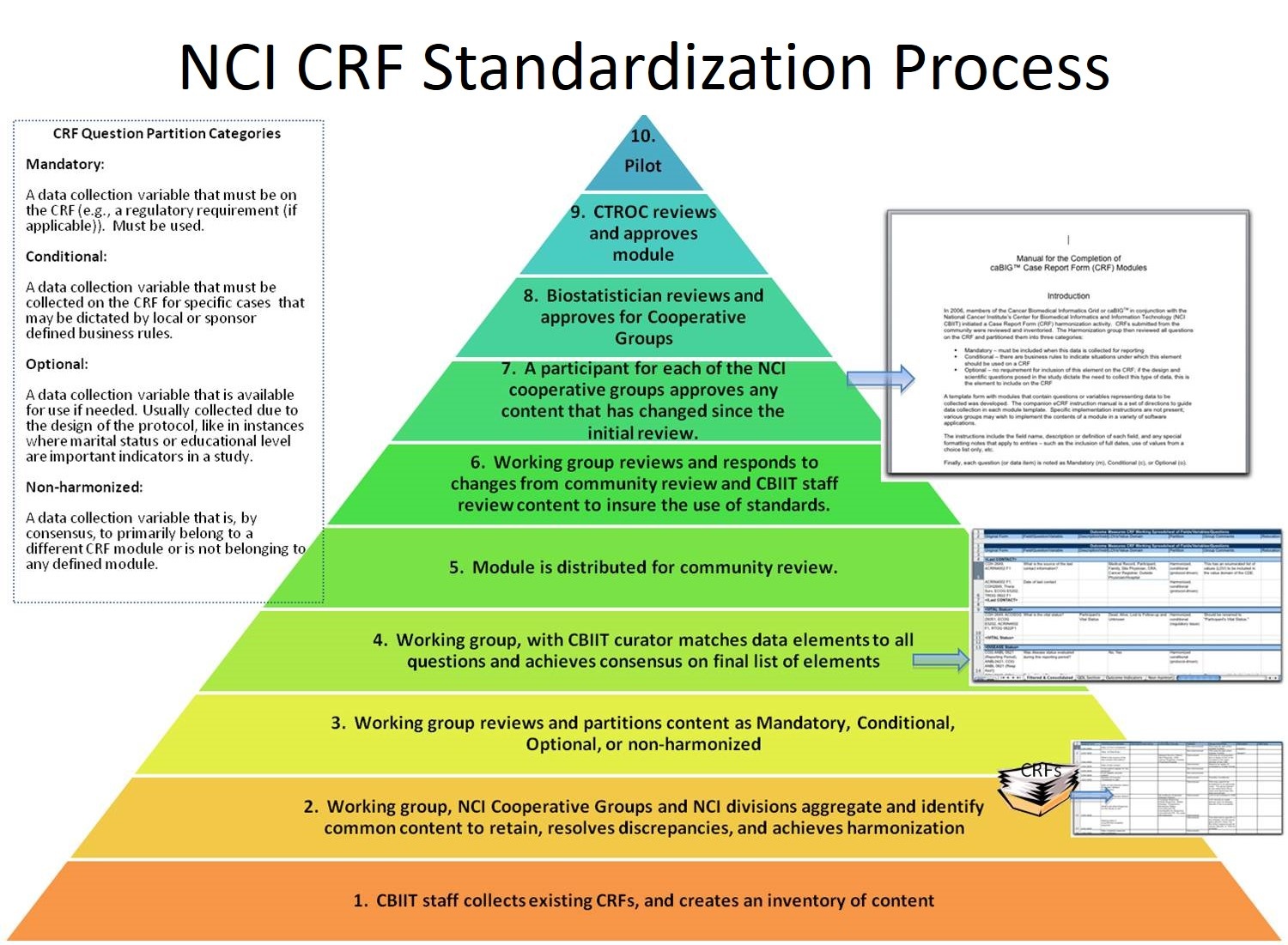
The Standardized Case Report Form (CRF) Work Group is focused on establishing a core library of harmonized and standardized Phase II and III CRFs and data collection modules through stakeholder consensus. A core library of harmonized and standardized CRFs will be highly valuable to the cancer clinical trials community because it will provide a standardized mechanism for comparing and aggregating information across the NCI's clinical trial portfolio. Additionally, a harmonized CRF library will improve the efficiency and accuracy of the routine review of safety, efficacy, and administrative data from ongoing NCI-funded clinical trials. Finally, by reducing the time spent in developing a data collection strategy per trial, this core library will allow for faster initiation of new trials; thus, speeding the process of delivering new and improved treatments to patients.
The Standardized CRF Work Group is part of the Study Conduct Special Interest Group (SIG) and the Clinical Trials Management Systems (CTMS) Workspace.
...
...
...
...
...
...
...
...
...
...
...
...
...
...
...
...
...
...
...
The general metrics from the Round 4 CRF Inventory include:
17 Contributing Organizations
Contributing Organizations: CCR, DCP, University of Pennsylvania, CDASH, NCCTG, CTMS Theradex, CIP (FIMSO), UCSD, CALGB, COH, ACOSOG, Duke, ACRIN, ECOG, University of Michigan, Baylor College of Medicine, and RTOG
237 Case Report Forms
7697 variables (total)
More metrics on the CRFs, variables and modules will be posted on this page and the Metrics Child Page: https://wiki.nci.nih.gov/display/CTMS/CRF+Project+Metrics
...