|
Page History
...
...
| Column | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| |||||||||
|
...
| width | 50% |
|---|
...
...
...
...
...
...
...
...
...
Under Review
...
...
...
Under Review
...
...
...
...
...
...
...
...
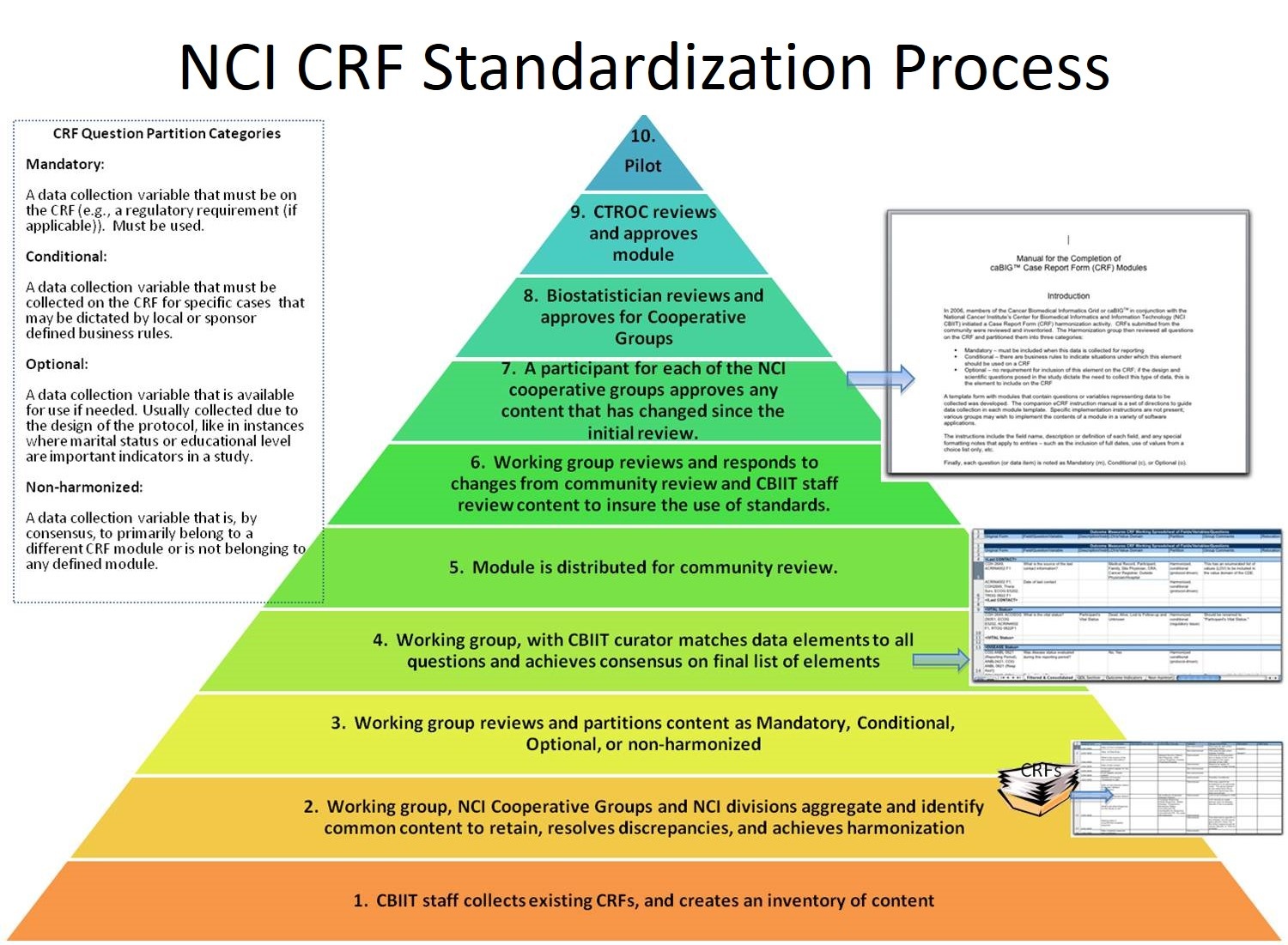
Harmonization/Standardization Process
...
...
...
...
...
...
The first Round of the CRF Harmonization and Standardization Project addressed a single module, Demography. This work began in June 2007, and the module has gone through the the entire process and has been approved by CTROC, and the CDEs have been made standard. The module is undergoing implementation/deployment for use in cancer clinical trials.
Round 2 included Adverse Events, Baseline Assessment, Participant Identification, Registration & Enrollment, and Protocol Deviations. Seven modules were completed from those 5 areas, which are Adverse Events, Medical History, Physical Exam, Participant Identification, Registration, Enrollment, and Protocol Deviations. These seven modules have been vetted by the Community and approved by CTROC. In the spring of 2010 the seven modules entered expanded community review. Community review completed three rounds of evaluation through 2010 and in March of 2011 was forwarded to NCI leadership for finalization.
Round 3 modules include Agents (looking at both standardizing vocabulary and the variables to be collected), Laboratory Tests/Results, Outcome Measures, and Staging/Extent of Disease. These modules have been reviewed by the Community, presented to CTROC, and are undergoing final review by specific groups in the cancer clinical trials community. It is anticipated that this set of content will be finalized in April of 2011.
The Round 4 modules: Header Information, Imaging/Radiology, Non-agent Study Interventions, Diagnosis/Pathology, Vital Signs, Eligibility Criteria, and Off Treatment/Off Study have completed the first round of harmonization in small workgroups. They will enter expanded community review upon the completion of Round 3 content, sometime in the spring of 2011.
Round 5 modules have not been identified at this point, but most likely will include Imaging variables, specifically Recist criteria. Additional content will be identified following a gap analysis once Round 4 work is complete.
...
The general metrics from the Round 4 CRF Inventory include:
17 Contributing Organizations
Contributing Organizations: CCR, DCP, University of Pennsylvania, CDASH, NCCTG, CTMS Theradex, CIP (FIMSO), UCSD, CALGB, COH, ACOSOG, Duke, ACRIN, ECOG, University of Michigan, Baylor College of Medicine, and RTOG
237 Case Report Forms
7697 variables (total)
More metrics on the CRFs, variables and modules will be posted on this page and the Project Metrics page.
CRF Project Wiki Child Pages
...