|
Page History
The Update Participating Sites section captures site statuses and dates. When updating sites, you must follow the the rules specified in Status Transition RulesInstitutional Review Board (IRB) section captures IRB contact information which is optional, however this information is required to upload a study from CTRP to ClinicalTrials.gov.
| Anchor | ||||
|---|---|---|---|---|
|
For Abbreviated trials, refer to Updating Participating Site Organization Records.
| Note | ||
|---|---|---|
| ||
The system validates the trial and site status transition(s) you make to each participating site when you review the trial information (as described in Reviewing and Submitting Trial Updates) before submitting the update. |
If you add a status that does not conform to the rules provided in Status Transition Rules, the system displays errors and/or warnings. Warnings indicate that fixing the record is optional. However, Errors indicate that you must correct the site status and/or status date.
How to Update Participating Site Status How to Update Institutional Review Board (IRB) Information
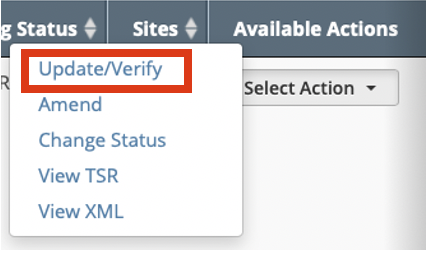
- Search for the trial whose participating site status IRB information you want to update. For instructions, refer to Searching for Trial Records. The Search Results table displays the results of your search and actions available (if any) for each record.
In the Available Actions column, click Select Action > Update/Verify. The Update Trial window lists the previously -recorded participating site statuses in the Participating Sites section.
Info Note: Only participating site(s) listed for a trial in CTRP can be updated using this mechanism. If the participating site has not been added to the trial, please contact CTRO to add the site to the trial.
Select new statuses and enter the corresponding trial status date(s) as necessary.
Click the Update/Verify button at the bottom of the page.
If you entered invalid values, error messages display instructions for correcting the statuses and/or dates.Fix any errors as necessary, and then click Update/Verify to resubmit.
recorded IRB
Select or enter the appropriate information in the text fields and drop-down lists. The following table describes the fields.
Once submitted successfully, a confirmation message will be displayed at the top of the pageField Label
Description/Instructions
Board Approval Number*
If the trial has been submitted and approved, type the number assigned to the protocol by the human subjects review board in the field. If the number does not appear in the IRB approval document, enter the date.
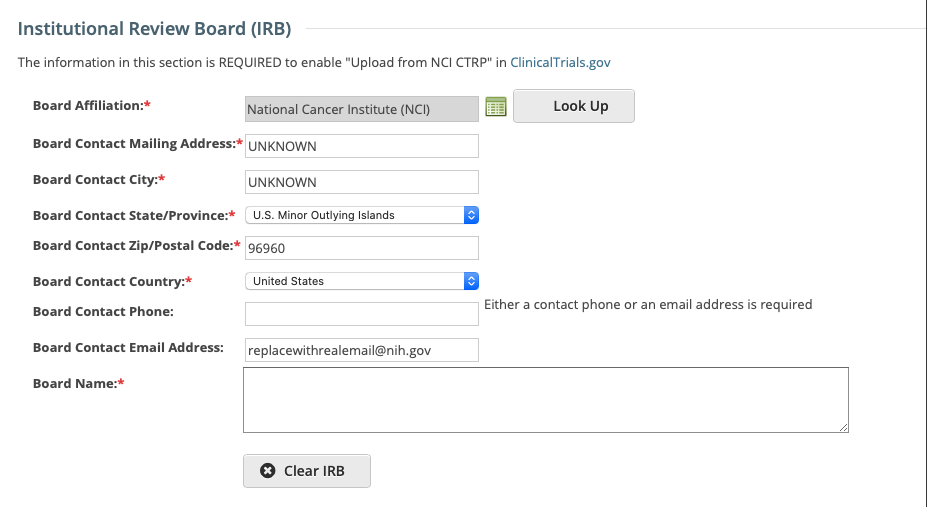
Board Affiliation*
Click Look Up to search for and select the Board name. If the name is not listed, register it with the CTRO. See Searching for Organizations.
When you select the Board name, the contact information is displayed automatically in the fields provided.
If you registered a new organization with the CTRO in the previous step, the system will notify you when the record is added so that you may continue your abstraction.
If applicable, complete any of the contact fields that are missing information.Board Name
The name of the affiliated organization.
Info Include Page Closure of Participating Sites When Trial Closes - Include v4.5 Closure of Participating Sites When Trial Closes - Include v4.5