|
Page History
| Multiexcerpt | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Since 1997, EVS has worked with many partners to build shared content and services, so that information can be effectively exchanged, interpreted and analyzed while minimizing overall effort and cost. Much of this content has been created and published using NCI Thesaurus and NCI Metathesaurus, as outlined in the following sections and described in more detail in the individual user profiles later, where other shared development efforts are also described.
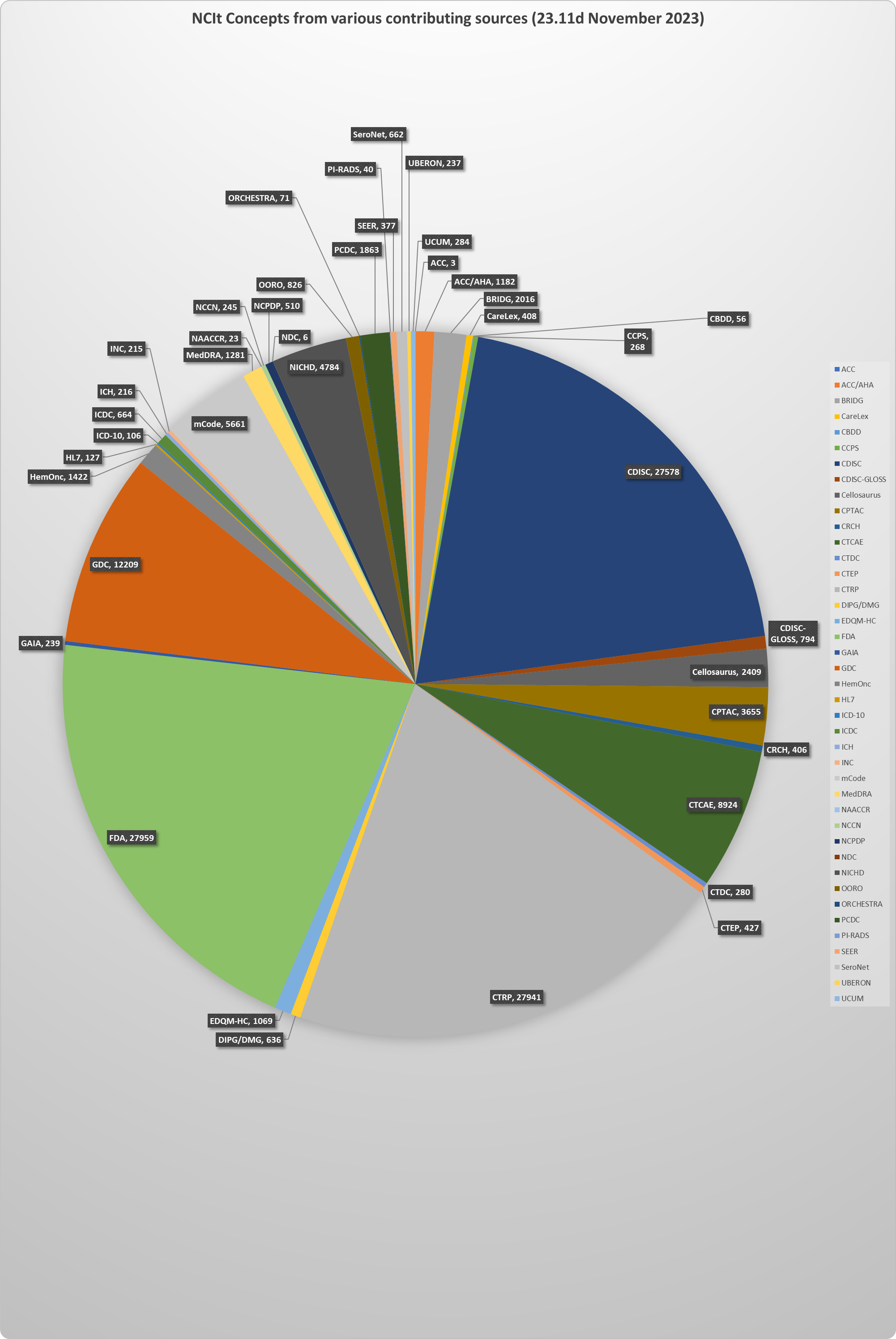
NCI Thesaurus (NCIt)NCI Thesaurus (NCIt) is NCI's core reference terminology. Over the last 16 years, it has also been adopted by FDA, CDISC, NCPDP and other partners as a shared standards development and coding environment, allowing participants to compare and harmonize with each other's content while taking advantage of full-text definitions, codes, and other features. About 125,000 out of the 178,080 current NCIt concepts have terms from both EVS and from one or more of these other sources. The chart below gives a detailed breakdown of the main areas of overlapping tagged content. Note that some partners have used NCI terms rather than tagging their own, so these figures understate the true extent of cross-source sharing. NCIt concepts with terms from tagged outside sources (23.11D: November 2023) NCIt concepts with tagged synonyms from each outside source (23.11D: November 2023)
NCI Metathesaurus (NCIm)NCI Metathesaurus (NCIm) was started in the late 1990s to gather up, translate between, and publish the many terminologies used by NCI, including some created in part or whole within NCI. Development of NCI Thesaurus started within NCIm as NewPDQ, an effort to extend and restructure the PDQ Terminology long used to code cancer clinical trial, research and public information resources. NCIm continues to provide a vital environment for EVS collaboration with NCI and other partners to develop, map and publish terminologies of shared interest, and responds to the requirements of a broad range of stakeholders within the cancer research and biomedical community. NCIt tagged content from outside sources is imported into NCIm with separate source tags. NCIt-derived sources are grouped separately below. The NCIm tags are mostly identical or very similar to those in NCIt; exceptions are CADSR, converted to NCI; HL7, converted to NCI-HL7; and CTRM, which is not imported. NCIm sources and the number of concepts to which each contributes (202302: May 2023)
Overall Counts (May 2023)
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Wiki Markup | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| {scrollbar:icons=false}
h1. {page-info:title}
{multi-excerpt:name=SharedTerminologyDevelopment}Since 1997, EVS has worked with many partners to build shared content and services, so that information can be effectively exchanged, interpreted and analyzed while minimizing overall effort and cost. Much of this content has been created and published using NCI Thesaurus and NCI Metathesaurus, as outlined below and described in more detail in the individual user profiles later, where other shared development efforts are also described.
{toc:minLevel=2}
h2. NCI Thesaurus (NCIt)
NCI Thesaurus (NCIt) is NCI's core reference terminology. Over the last 10 years, it has also been adopted by FDA, CDISC, NCPDP and other partners as a shared standards development and coding environment, allowing participants to compare and harmonize with each other's content while taking advantage of full-text definitions, codes, and other features.
About 40,000 out of the nearly 100,000 current NCIt concepts have terms from both EVS and from one or more of these other sources. The chart below gives the last available detailed breakdown, for June 2010. Note that some partners have used NCI terms rather than tagging their own, so these figures understate the true extent of cross-source sharing.
{color:#002060}{*}NCIt concepts with terms from tagged outside sources (10.06e: June 2010)*{color}
!worddavd211bdc8d8a00c934a783d9a6e2db9d8.png|height=508,width=629,alt=""!
h2. NCI Metathesaurus (NCIm)
NCI Metathesaurus (NCIm) ... \[text pending\]{multi-excerpt}
{scrollbar:icons=false} |