This section describes how to abstract and edit the scientific details of clinical trials.
About Scientific Data
- Trial descriptions
- Trial design, including design details, outcome measures, and eligibility criteria
- Diseases and conditions
- Data Table 4 anatomic sites
- Biomarkers (Markers)
- Interventions
- Arms for Interventional trials
- Groups and cohorts for Non-interventional trials
- Sub-groups
After a trial has been accepted, if you log in with an appropriate role, the Scientific Data menu contains an option for each of the above categories.
The trial summary remains displayed at the top of each page as you navigate through the Scientific Data section.
When you have completed abstracting all trial information on a given page, be sure to click the Save button at the bottom of each page before you proceed to another task.
Abstracting Trial Descriptions
- Brief title
- Brief summary
Additionally, they may include a detailed description.
How to Abstract Trial Descriptions
Search for the trial of interest. For instructions, refer to Searching for Trials in PA.
In the search results, click the NCI Trial Identifier link for that trial. The Trial Identification page appears.
On the Trial Identification page, check out the trial. For instructions, refer to Checking In and Checking Out Trials. (This checkout step is optional for Super Abstractors.)
On the Scientific Data menu, click Trial Description. The Trial Description page appears.
On Trial Description page, in the various fields, specify the appropriate information. The following table describes the fields. An asterisk (*) indicates a required field.
You can expand text fields and table cells to reveal all of their content
To increase the size of a text field or table cell, click and drag the lower right corner handle.
Trial Description
Instruction
Brief Title*
Enter a title that summarizes the purpose of the trial.
Brief Summary*
Enter a summary description of the trial.
Detailed Description
Optionally, enter all the information that accurately describes the trial in detail.
- Click Save.
Abstracting Interventional Trial Design
Abstracting Non-Interventional Trial Design
Abstracting Outcomes
Abstracting Eligibility Criteria
Abstracting Diseases and Conditions
Abstracting Data Table 4 Anatomic Sites
For instructions, refer to the following pages:
Abstracting Biomarkers
For instructions, refer to the following pages:
Abstracting Interventions
Additionally, Protocol Abstraction provides a structure for describing drug, radiation, and surgical procedure details. It generates a description from values you provide for such parameters as dose, target site, frequency of administration, etc. Obsolete terms Some intervention terms become obsolete over time. When you validate the trial, the system displays an error message if the abstraction contains an obsolete term. If this occurs, look up the intervention name and replace the obsolete term with the new one. Trials can have multiple intervention records. Each record displays the following information about the intervention: Intervention data definitions Column Name Definition Name Standard name used to refer to each intervention. For drugs, this is the generic name. For investigational new drugs that do not yet have a generic name, this may be the chemical name, company code, or serial number. Other Names Synonym or other name
1
Description For drug interventions, a summary of the drug’s chemical nature and (potential) mechanism of action, and/or details such as dosage form, dosage, frequency, and duration. Tip You can generate descriptions for drug, radiation, and surgical procedure interventions using Protocol Abstraction’s structured approach. To do so, see Generating Drug, Radiation, and Procedure/Surgery Descriptions. Type Mode of intervention, for example, drug or device. Additionally, you can add, edit, reorder, and delete records. Sorting intervention records You can change the sort order (ascending/descending) of any column other than Edit and Delete by clicking the column name and selecting the up or down arrow that appears. For instructions, refer to the following pages:
For other interventions, a summary of the procedure or device that provides key details that distinguish it from similar interventions.
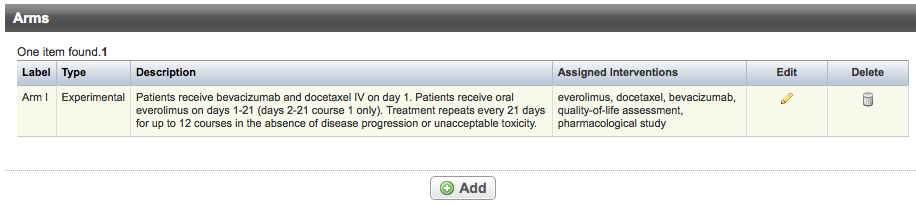
Abstracting Arms
When adding arms to a trial, you assign to them the interventions currently abstracted for the trial. For instructions on abstracting interventions, refer to Abstracting Interventions.
The following rules apply to each trial:
There must be at least one arm/group that implies that the entire study subject population receives the same intervention.
The number of arms/groups added to the trial must coincide with the number indicated in the trial’s Design Details. See Abstracting Interventional Trial Design Details.
- Every arm must have at least one assigned intervention (except for the arm type "no intervention").
- Every intervention must be assigned to at least one arm.
- There must be only one arm with the same combination of description and associated intervention.
- Arm details for single-arm trials are not required.
Trials can have multiple arm records. Each record displays the following arm information:
Field | Definition |
|---|---|
Label | Short name used to identify the arm or comparison group. |
Type | Function of the arm. Valid types are as follows:
|
Description | Brief description of the arm or comparison group to distinguish it from other arms/groups in the trial. |
Assigned Interventions | Intervention(s) assigned to a given arm. 1 |
____
- Interventions imported from ClinicalTrials.gov may not match CTRP terminology. ↩
For instructions, refer to the following pages:
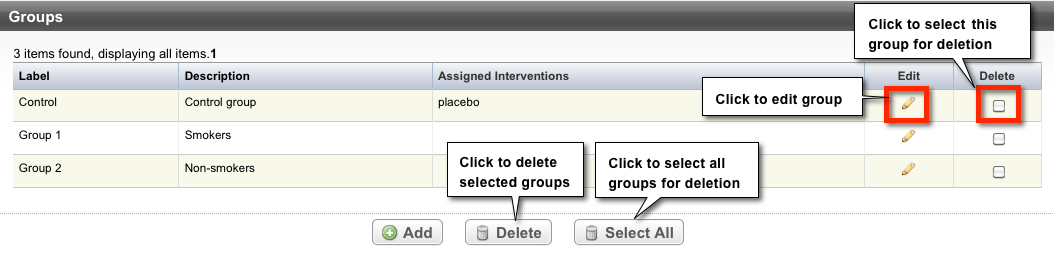
Abstracting Groups and Cohorts
When adding groups to a trial, you assign to them the interventions currently abstracted for the trial. For instructions on abstracting interventions, refer to Abstracting Interventions.
The following rules apply to each trial:
There must be at least one group/cohort that implies that the entire study subject population receives the same intervention.
The number of groups/cohorts added to the trial must coincide with the number indicated in the trial’s Design Details. See Abstracting Non-Interventional Trial Design Details.
- Every group/cohort must have at least one assigned intervention.
- Every intervention must be assigned to at least one group/cohort.
- A trial must have only one group with the same combination of description and associated intervention.
Trials can have multiple group/cohort records. Each record displays the following group information:
| Field | Definition |
|---|---|
Label | Short name used to identify the group |
Description | Explanation of the nature of the study group (e.g., those with a condition and those without a condition; those with an exposure and those without an exposure). |
Assigned Interventions | Intervention assigned to a given group |
Group Records
For instructions, refer to the following pages:
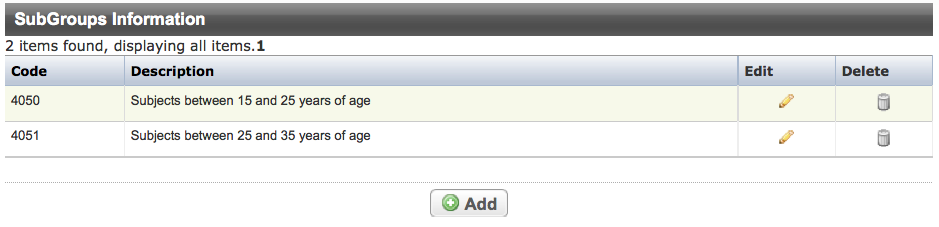
Abstracting Sub-Groups
Trials can have multiple sub-group records. Each record displays the following information about the disease or conditions:
Field | Definition |
|---|---|
Code | Characters, or phrase used to identify the sub-group |
Description | Stratification criteria |
Sub-Groups Page
For more instructions, refer to the following pages: