CTRP Accrual site enables you to review the collection of non PII (Personally Identifiable Information) study subject data associated with a particular non-industrial trial registered with the CTRP.
How to Review Study Subject Records
- Select the trial you want to work with by following instructions in Selecting Listed Trials in Accrual or Searching for Trials in Accrual , and clicking the corresponding NCI Trial Identifier link.
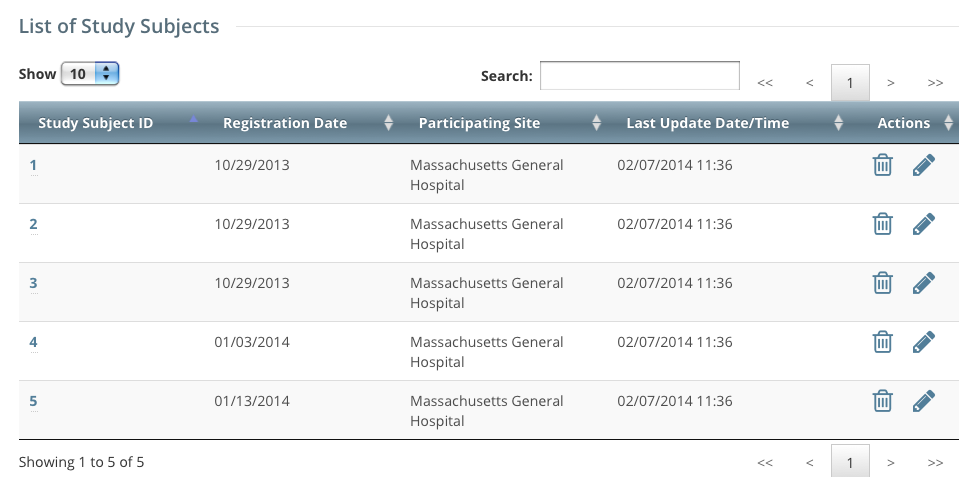
The Accrual Submissions page displays any accrual records that may have been submitted previously.
- Click the Study Subject ID of interest.
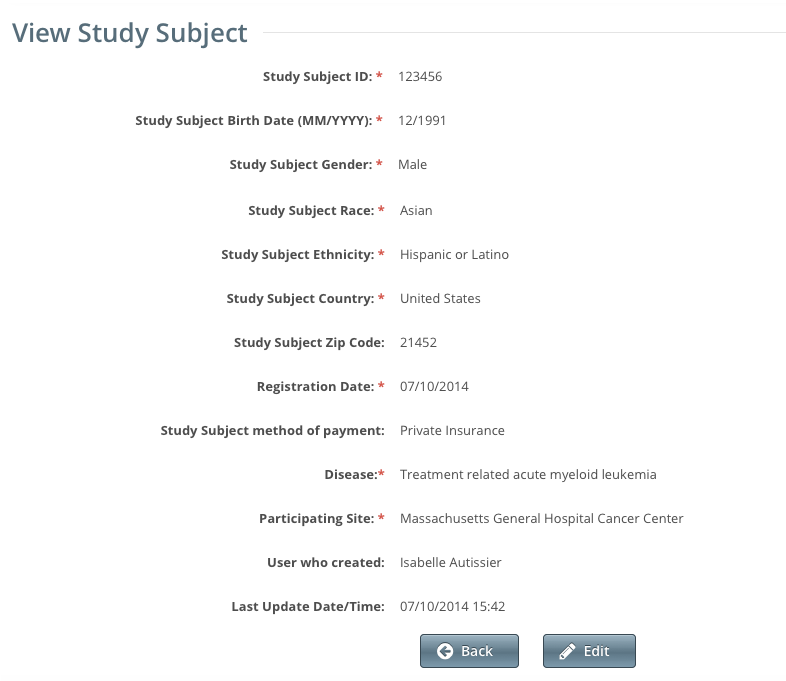
The Study Subject page displays the study subject's demographic details recorded to date as well as information about the record itself.
- To return to the accrual record, click Back.
- To change the study subject record, click Edit, and follow the instructions in Adding Study Subjects.