This section provides instructions for recording the progress of a trial along its processing life cycle.
About Milestones
Milestones are used to record the progress of a trial along its processing life cycle and to establish the processing status. The system establishes some milestones at specific events during the process. For example, it sets the trial processing status to "Submitted" for a trial that has not yet been validated, and to "Accepted" once the trial has been validated and accepted.
The system records milestones as sets by submission number. For example, an original trial has one set of milestones (one submission), but an amended trial may have two or more submissions. Each subsequent submission has its own set of recorded milestones.
About Milestones
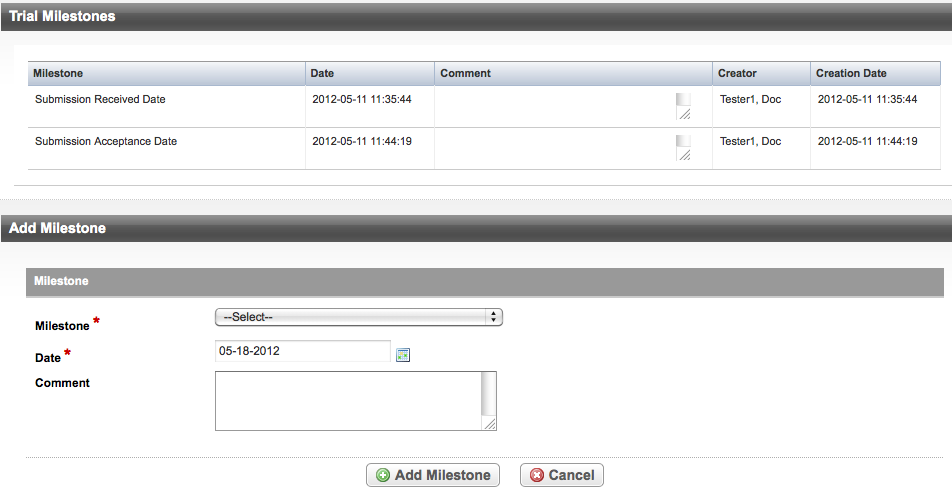
The system records milestones as sets by submission number. For example, an original trial has one set of milestones (one submission), but an amended trial may have two or more submissions. Each subsequent submission has its own set of recorded milestones. The figure below illustrates the trial milestones for an accepted trial.
The figure below illustrates the trial milestones for an accepted trial.
Viewing and Adding Milestones
Because milestones indicate the progress of a trial, you can view, but not change recorded milestones. However, you can add records throughout the life cycle of the trial submission. You can not add milestones to trials that are currently on hold.
For trials with one or more amendments, the system displays milestones in sets, one for each submission. You can add milestones only when the Submission Number is the Active Milestone set, as shown below.
If you select an Inactive Submission set, that is, one that contains a previous submission, you can not add new milestones. In the figure below, the selected Submission Number is 5 (out of 6 submissions).
An abstractor can reject an amendment submission without rejecting the whole trial.
If an abstractor has rejected an amendment submission, the system displays the last Active milestone set by default, as shown below.
How to View and Add Milestones
- In any list of trial search results, click the NCI Trial Identifier link for the trial of interest.
- On the Trial Overview menu, click Trial Milestones. The Trial Milestones page appears and displays all milestones recorded to date.
- To add a milestone, in the Add Milestone section at the bottom of the page, select the milestone you want to add from the drop-down list. The following sections list valid milestones, processing statuses, and the rules for recording them. For milestone definitions, see Trial milestone definitions.
- In the Date field, enter the milestone date. The system enters the current date by default.
In the Comment field, type the action that triggered the milestone, or any comments about the event. The comments field is limited to 200 characters.
- Click Add Milestone.
If you are adding Late Rejection milestone to an amended trial, the system gives you a choice.
- If this message appears, select Reject Entire Trial or select Reject This Amendment Only.
The system displays the milestone at the bottom of the list of milestones in the Trial Milestones section of the page. - Repeat the steps above after you complete each abstraction update as appropriate.
Un-rejecting a Trial
A Super Abstractor can un-reject a trial that has a processing status of Rejected.
How to Un-reject a Trial
- In any list of trial search results, click the NCI Trial Identifier link for the trial of interest.
- On the Trial Overview menu, click Trial Milestones. The Trial Milestones page appears and displays all milestones recorded to date.
- In the Add Milestone section at the bottom of the page, click Un-Reject Trial. The system prompts you to provide a comment.
- Type a comment and click OK. The system appends your comment (along with a date/time-stamp and your user name) to the comments for the previously active milestone. The system displays information about this un-reject operation on the Trial Milestones page and on the Submissions tab of the Trial History Information page.
Milestone Recording Rules
The following rules determine the order in which you can record the milestones:
- You can record most milestones multiple times per trial, following the order of the trial submission processing life cycle. However, you can add the following milestones only once per trial submission:
- Submission Received Date
- Submission Rejection Date
- Initial Abstraction Verified Date
- Initial Submission to ClinicalTrials.gov
- You cannot record milestones with a future date.
- Each milestone date must follow the preceding milestone date.
If the most current processing status is this... | You can record this milestone |
|---|---|
Submitted | Submission Received Date |
Accepted and all Administrative and Scientific abstraction activities have been completed | QC Completed Date. Recording this milestone sets the trial processing status to Abstracted and Trial Verification Date to the date that you record this milestone. |
Verification Pending and a Submitter TSR Feedback Date milestone has been recorded | Initial Abstraction Verified Date. |
Verification Pending and a Submitter TSR Feedback Date milestone has not been recorded | Initial Abstraction Verified Date. |
Abstraction Verified Response | On-going Abstraction Verified Date |
Abstraction Verified No Response | On-going Abstraction Verified Date |
Abstracted | Trial Summary Report Date. |
Submission Terminated | Submission Reactivated Date. This is the only milestone that you can record after the Submission Terminated Date. |
If the most current processing status is this... | You can not record this milestone |
|---|---|
Submitted | Trial Summary Report Date |
Accepted | Trial Summary Report Date |
This date... | Must precede this date |
|---|---|
Ready for Administrative QC Date | Administrative QC Start Date |
Ready for Scientific QC Date | Scientific QC Start Date |
Administrative QC Start Date | Administrative QC Completion Date |
Scientific QC Start Date | Scientific QC Completion Date |
TSR Date | Submitter TSR Feedback Date |
Administrative Processing Start Date | Administrative Processing Completed Date |
Scientific Processing Start Date | Scientific Processing Completed Date |
Submission Terminated Date | Submission Reactivated Date |
You can not record this milestone... | If this is true |
|---|---|
QC Start Date |
|
Trial Summary Report Date |
|
Submitter Trial Summary Report Feedback Date |
|
Initial Abstraction Verified Date |
|
Initial Submission to ClinicalTrials.gov |
|
On-going Abstraction Verified Date |
|
Late Rejection Date |
|
Ready for QC Date |
|
Submission Terminated Date | The trial has been rejected |
Milestone Definitions
Milestone | Definition |
|---|---|
Submission Received Date | Date the trial was successfully submitted via the NCI CTRP Registration Site |
Submission Acceptance Date | Date the document specialist accepted the trial submission for validation |
Submission Rejection Date | Date the document specialist rejected the trial submission during validation |
Administrative Processing Start Date | Date of initial abstraction of administrative details |
Administrative Processing Completed Date | Date the abstraction of administrative details was completed |
Scientific Processing Start Date | Date of initial abstraction of the scientific details |
Scientific Processing Completed Date | Date the abstraction of scientific details was completed |
Ready for Administrative QC Date | Date the trial abstraction of administrative data was completed and available for Quality Control (QC) of the administrative data |
Ready for Scientific QC Date | Date the trial abstraction of scientific data was completed and available for Quality Control (QC) of the scientific data |
Administrative QC Start Date | Date of initial QC of administrative data |
Scientific QC Start Date | Date of initial QC of scientific data |
Administrative QC Start Date | Date of initial QC of the administrative data |
Scientific QC Start Date | Date of initial QC of the scientific data |
Administrative QC Completed Date | Date that QC of the administrative data was completed |
Scientific QC Completed Date | Date that QC of the scientific data was completed |
Ready for Trial Summary Report Date | Date the abstraction was completed. The system adds this milestone automatically. |
Trial Summary Report Date | Date the Trial Summary Report was sent to the principal investigator or trial submitter. The system adds this milestone automatically. Special treatment of TSRs for Abbreviated Trials The system does not send Trial Summary Reports for Abbreviated Trials. You can download the TSRs for these trials from the Trial Related Documents page as needed. |
Submitter Trial Summary Report Feedback Date | Date input was received by a document specialist from a principal investigator or trial submitter regarding data contained on the Trial Summary Report |
Initial Abstraction Verified Date | Date the document specialist recorded that the abstracted data was verified upon receipt of the submitter's TSR feedback. Equal to the TSR Sent Date plus six business days if the most current processing status is Abstracted. TSR feedback is due within five business days following the TSR Sent Date. The Initial Abstraction Verified Date can be later if no feedback is received. (A business day is any weekday that is not a Federal holiday. For a list of Federal holidays, refer to the U.S. Office of Personnel Management's list of Federal Holidays.) |
On-going Abstraction Verified Date | Date the document specialist recorded that abstracted data was verified, following a post-verified status when there has been an abstraction update or a delay in the submitter's feedback |
Late Rejection Date | Date the document specialist rejected the trial submission at any time after initial trial acceptance 1 |
Submission Terminated Date | Date the document specialist stopped the trial submission process |
Submission Reactivated Date | Date the document specialist resumed the trial submission process |
- The Late Rejection Date applies to both original and amendment submissions. ↩
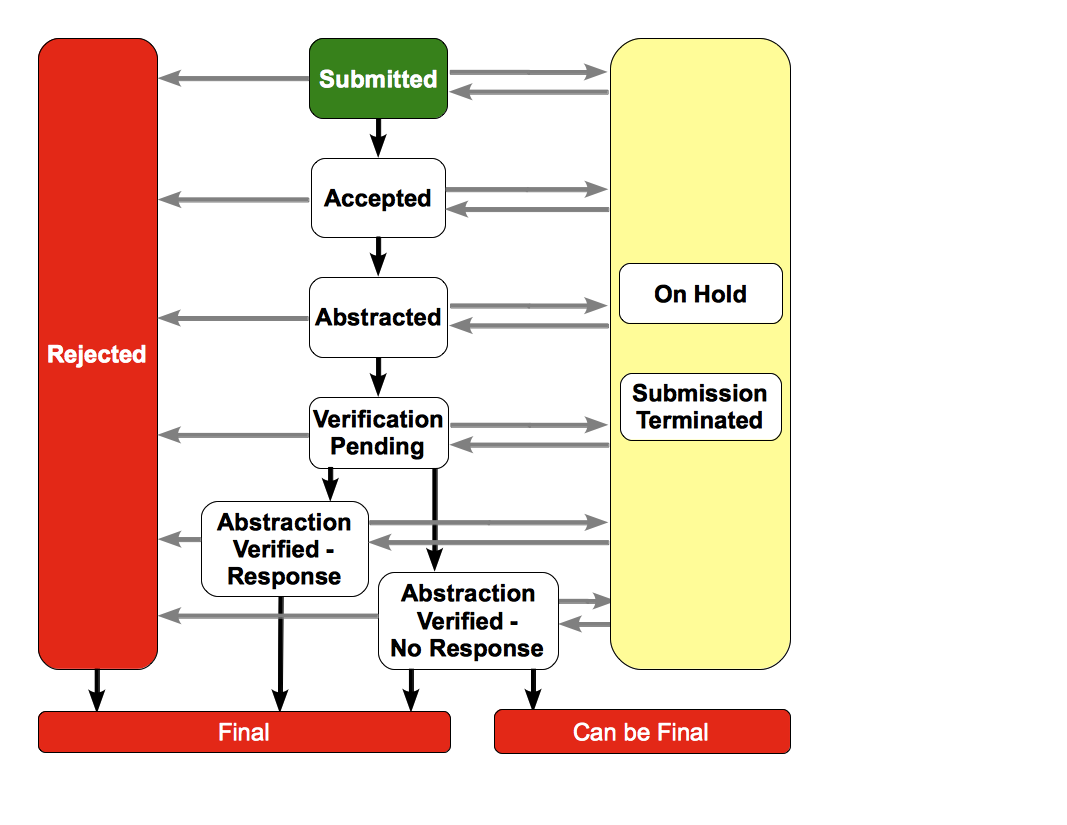
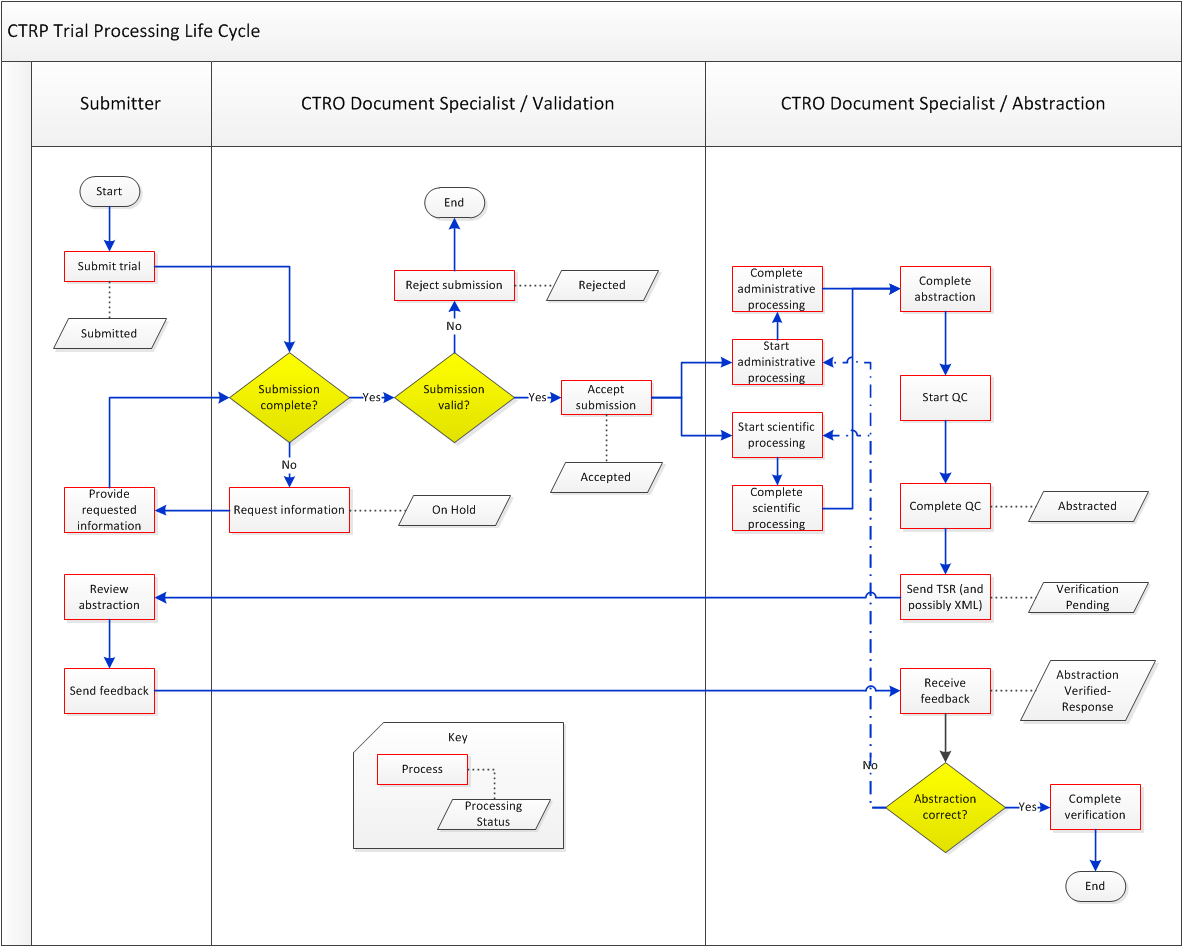
Trial Processing Life Cycle
The following diagram illustrates the typical life cycle of a trial and the relationship between actions and processing statuses:
Trial Summary Report Life Cycle
The system generates TSRs upon the document specialist’s request via the trial search functionality, via the View TSR option on the Trial Overview menu, and via abstraction validation. TSRs are also generated and emailed to the submitter when the Trial Summary Report Sent Date milestone is recorded.
TSR feedback from the submitter or principal investigator triggers milestone events in the system. The table below shows the relationships between processing life cycle, milestone events, and TSRs.
Life Cycle Phase/Milestone | TSR Sent From | To Recipient |
|---|---|---|
Send TSR/TSR Sent Date | CTRO |
|
Receive TSR feedback/Submitter TSR Feedback Date | Submitter (with feedback) | CTRO |
Trial amendments follow a similar system of reporting and feedback as the original submission, though the TSR includes additional amendment-related data.