Since 1997, EVS has worked with many partners to build shared content and services, so that information can be effectively exchanged, interpreted and analyzed while minimizing overall effort and cost. Much of this content has been created and published using NCI Thesaurus and NCI Metathesaurus, as outlined in the following sections and described in more detail in the individual user profiles later, where other shared development efforts are also described.
NCI Thesaurus (NCIt)
NCI Thesaurus (NCIt) is NCI's core reference terminology. Over the last 16 years, it has also been adopted by FDA, CDISC, NCPDP and other partners as a shared standards development and coding environment, allowing participants to compare and harmonize with each other's content while taking advantage of full-text definitions, codes, and other features.
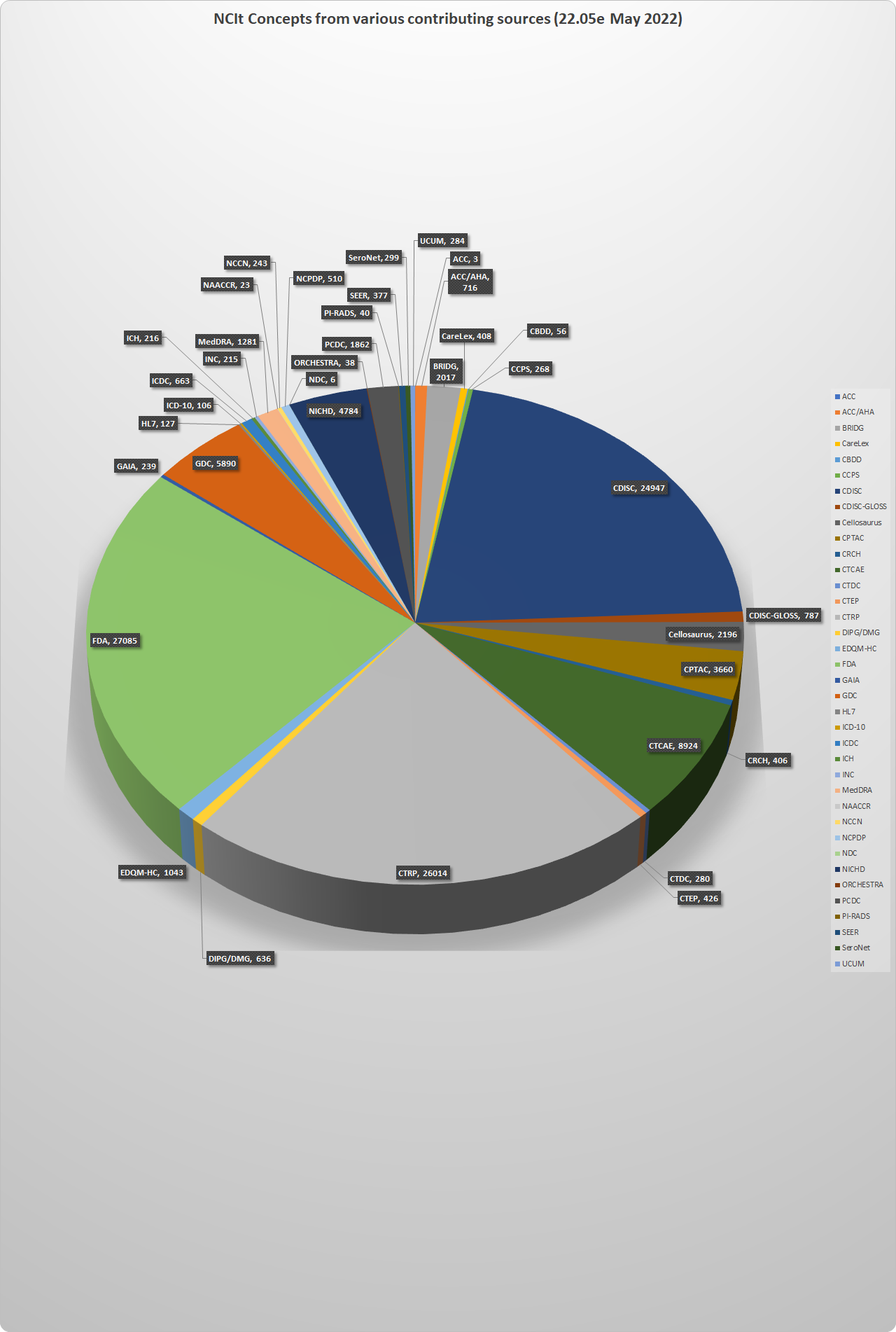
About 117,000 out of the 154,946 current NCIt concepts have terms from both EVS and from one or more of these other sources. The chart below gives a detailed breakdown of the main areas of overlapping tagged content. Note that some partners have used NCI terms rather than tagging their own, so these figures understate the true extent of cross-source sharing.
NCIt concepts with terms from tagged outside sources (22.05e: May 2022)
NCIt concepts with tagged synonyms from each outside source (22.05e: May 2022)
NCIt Concepts | Source |
|---|---|
| 3 | ACC |
| 716 | ACC/AHA |
2017 | BRIDG |
| 408 | CareLex |
| 56 | CBDD |
268 | CCPS |
| 24947 | CDISC |
| 787 | CDISC-GLOSS |
| 2196 | Cellosaurus |
| 3660 | CPTAC |
| 406 | CRCH |
| 8924 | CTCAE |
| 280 | CTDC |
| 426 | CTEP |
| 26014 | CTRP |
| 636 | DIPG/DMG |
| 1043 | EDQM-HC |
| 27085 | FDA |
| 239 | GAIA |
| 5890 | GDC |
| 127 | HL7 |
| 106 | ICD-10 |
| 663 | ICDC |
| 216 | ICH |
| 215 | INC |
| 1281 | MedDRA |
| 23 | NAACCR |
| 243 | NCCN |
| 510 | NCPDP |
| 6 | NDC |
| 4784 | NICHD |
| 38 | ORCHESTRA |
| 1862 | PCDC |
| 40 | PI-RADS |
| 377 | SEER |
| 299 | SeroNet |
| 284 | UCUM |
NCI Metathesaurus (NCIm)
NCI Metathesaurus (NCIm) was started in the late 1990s to gather up, translate between, and publish the many terminologies used by NCI, including some created in part or whole within NCI. Development of NCI Thesaurus started within NCIm as NewPDQ, an effort to extend and restructure the PDQ Terminology long used to code cancer clinical trial, research and public information resources. NCIm continues to provide a vital environment for EVS collaboration with NCI and other partners to develop, map and publish terminologies of shared interest, and responds to the requirements of a broad range of stakeholders within the cancer research and biomedical community.
NCIt tagged content from outside sources is imported into NCIm with separate source tags. NCIt-derived sources are grouped separately below. The NCIm tags are mostly identical or very similar to those in NCIt; exceptions are CADSR, converted to NCI; HL7, converted to NCI-HL7; and CTRM, which is not imported.
NCIm sources and the number of concepts to which each contributes (202105: May 2021)
NCIm concepts | Source Label - NCIt-derived | Source Name |
|---|---|---|
| 713 | ACC-AHA | American College of Cardiology/American Heart Association Clinical Data Terminology, 2021_01D |
| 334 | BioC | BioCarta online maps of molecular pathways, adapted for NCI use, 2021_01D |
| 27 | BRIDG | Biomedical Research Integrated Domain Group Model, 2021_01D |
| 1148 | BRIDG_3_0_3 | Biomedical Research Integrated Domain Group Model, 3.0.3, 2021_01D |
| 1658 | BRIDG_5_3 | Biomedical Research Integrated Domain Group Model, 5.3, 2021_01D |
| 17974 | caDSR | Cancer Data Standards Registry and Repository, 2021_01D |
| 407 | CARELEX | Content Archive Resource Exchange Lexicon, 2021_01D |
| 53 | CBDD | Chemical Biology and Drug Development Vocabulary, 2021_01D |
| 265 | CCPS | Childhood Cancer Predisposition Study, 2022_01E |
| 923 | CDC | U.S. Centers for Disease Control and Prevention, 2021_01D |
| 26120 | CDISC | Clinical Data Interchange Standards Consortium, 2021_01D |
| 788 | CDISC-GLOSS | Clinical Data Interchange Standards Consortium Glossary Terms, 2021_01D |
2167 | CELLOSAURUS | Cellosaurus, 2021_01D |
| 3688 | CPTAC | Clinical Proteomic Tumor Analysis Consortium, 2021_01D |
| 406 | CRCH | Cancer Research Center of Hawaii Nutrition Terminology, 2021_01D |
| 775 | CTCAE | Common Terminology Criteria for Adverse Events, 2021_01D |
| 5599 | CTCAE_3 | Common Terminology Criteria for Adverse Events 3.0, 2021_01D |
| 4022 | CTCAE_5 | Common Terminology Criteria for Adverse Events 5.0, 2021_01D |
| 276 | CTDC | Clinical Trial Data Commons, 2021_01D |
| 318 | CTEP | Cancer Therapy Evaluation Program, 2004 |
| 373 | CTEP-SDC | Cancer Therapy Evaluation Program - Simple Disease Classification, 2021_01D |
| 25582 | CTRP | Clinical Trials Reporting Program, 2021_01D |
| 903 | DCP | NCI Division of Cancer Prevention Program, 2021_01D |
| 114 | DICOM | Digital Imaging Communications in Medicine, 2021_01D |
| 627 | DIPG_DMG | Diffuse Intrinsic Pontine Glioma/Diffuse Midline Glioma Terminology, 2022_01E |
| 694 | DTP | NCI Developmental Therapeutics Program, 2021_01D |
| 1041 | EDQM-HC | European Directorate for the Quality of Medicines & Healthcare, 2021_01D |
| 25994 | FDA | U.S. Food and Drug Administration, 2021_01D |
| 207 | GAIA | Global Alignment of Immunization Safety Assessment in pregnancy, 2021_01D |
| 5472 | GDC | NCI Genomic Data Commons, 2021_01D |
| 281 | GENC | Geopolitical Entities, Names, and Codes Standard, 2021_01D |
| 106 | ICD-10 | International Classification of Diseases, 10th Edition, 2022_01E |
| 671 | ICDC | NCI Integrated Canine Data Commons, 2021_01D |
| 215 | ICH | International Conference on Harmonization, 2021_01D |
| 215 | INC | International Neonatal Consortium, 2021_01D |
| 156 | JAX | Jackson Laboratories Mouse Terminology, adapted for NCI use, 2021_01D |
| 244 | KEGG | KEGG Pathway Database, 2021_01D |
| 166401 | NCI | National Cancer Institute Thesaurus, 2021_01D |
| 5078 | NCI-GLOSS | NCI Dictionary of Cancer Terms, 2021_01D |
| 5392 | NCI-HGNC | NCI HUGO Gene Nomenclature Committee, 2021_01D |
| 129 | NCI-HL7 | NCI Health Level 7, 2021_01D |
| 509 | NCPDP | National Council for Prescription Drug Programs, 2021_01D |
| 5 | NDC | National Drug Code, 2021_01D |
| 4782 | NICHD | National Institute of Child Health and Human Development, 2021_01D |
| 1200 | PCDC | Pediatric Cancer Data Commons, 2021_01D |
| 39 | PI-RADS | Prostate Imaging Reporting and Data System, 2021_01D |
| 169 | PID | National Cancer Institute Nature Pathway Interaction Database, 2021_01D |
| 310 | RENI | Registry Nomenclature Information System, 2021_01D |
| 296 | SeroNet | Serological Sciences Network Terminology, 2022_01E |
| 1456 | UCUM | Unified Code for Units of Measure, 2021_01D |
| 20 | WHO | World Health Organization (WHO) Terminology, 2022_01E |
| 25 | ZFIN | Zebrafish Model Organism Database, 2021_01D |
NCIm concepts | Source Label - Other | Source Name |
|---|---|---|
| 15882 | AOD | Alcohol and Other Drug Thesaurus, 2000 |
| 276 | AOT | Authorized Osteopathic Thesaurus, 2003 |
| 13257 | CBO | Clinical Bioinformatics Ontology, 2011_02 |
| 541 | CCSR_ICD10CM | Clinical Classifications Software Refined for ICD-10-CM, 2021 |
| 356 | CCSR_ICD10PCS | Clinical Classifications Software Refined for ICD-10-PCS, 2021 |
| 962 | CDCREC | Race & Ethnicity - CDC, 1.2 |
| 3078 | COSTAR | COSTAR, 1989-1995 |
| 16494 | CSP | CRISP Thesaurus, 2006 |
| 3843 | CST | COSTART, 1995 |
| 318 | CTEP | Cancer Therapy Evaluation Program, 2004 |
| 529 | CVX | Vaccines Administered, 2017_02_08, 2020_07_28 |
| 9620 | DRUGBANK | DrugBank, 5.0_2016_06_22, 5.0_2020_07_30 |
| 7289 | DXP | DXplain, 1994 |
| 102562 | FMA | Foundational Model of Anatomy Ontology, 4_15 |
| 7495 | GARD | Genetic and Rare Diseases Information Center, 2015_04 |
| 71208 | GO | Gene Ontology, 2020_05_02 |
| 7591 | HCPCS | Healthcare Common Procedure Coding System, 2020 |
| 42505 | HGNC | HUGO Gene Nomenclature Committee, 2020_05 |
| 9061 | HL7V3.0 | HL7 Vocabulary Version 3.0, 2019_12 |
| 17747 | HPO | Human Phenotype Ontology, 2020_06_08 |
| 17424 | ICD10 | ICD10, 2016 |
| 103391 | ICD10CM | International Classification of Diseases, 10th Edition, Clinical Modification, 2021 |
| 190986 | ICD10PCS | ICD-10-PCS, 2021 |
| 20937 | ICD9CM | International Classification of Diseases, Ninth Revision, Clinical Modification, 2014 |
| 2874 | ICDO | International Classification of Disease for Oncology, 3_2 |
| 748 | ICPC | International Classification of Primary Care, 1993 |
| 5055 | ISO3166-2 | International Organization for Standardization 3166-2, 2012 |
| 167298 | LNC | LOINC, 268 |
| 41 | MCM | McMaster University Epidemiology Terms, 1992 |
| 332 | MDBCAC | Mitelman Database of Chromosome Aberrations in Cancer, 2005_12 |
| 59238 | MDR | Medical Dictionary for Regulatory Activities Terminology (MedDRA), 23_1 |
| 3431 | MED-RT | Medication Reference Terminology, 2020_09_08 |
| 2109 | MEDLINEPLUS | MedlinePlus Health Topics, 20191029 |
| 900 | MGED | MGED Ontology, 131 |
| 449367 | MSH | Medical Subject Headings, 2021_2020_07_16 |
| 212203 | MTH | UMLS Metathesaurus, 2020AB |
| 11 | MTHCMSFRF | Metathesaurus CMS Formulary Reference File, MTHCMSFRF_2014 |
| 19009 | MTHICD9 | International Classification of Diseases, Ninth Revision, Clinical Modification, Metathesaurus additional entry terms, 2014 |
| 1633 | MTHMST | Metathesaurus Version of Minimal Standard Terminology Digestive Endoscopy, 2001 |
| 69394 | MTHSPL | Metathesaurus FDA Structured Product Labels, 2020_08_28 |
| 84 | MVX | Manufacturers of Vaccines, 2020_09_04 |
| 1983453 | NCBI | NCBI Taxonomy, 2020_05_21 |
| 7984 | NCIMTH | NCIMTH |
| 0 | NCISEER | NCI SEER ICD Neoplasm Code Mappings, 1999 |
| 1887 | NPO | NPO: NanoParticle Ontology for Cancer Nanotechnology Research, 2011_12_08 |
| 97276 | OMIM | Online Mendelian Inheritance in Man, 2020_07_27 |
| 12398 | PDQ | Physician Data Query, 2016_07_31 |
| 2039 | PMA | Portfolio Management Application, 2010 |
| 260 | PNDS | Perioperative Nursing Data Set, 4_2018 |
| 940 | QMR | Quick Medical Reference (QMR), 1996 |
| 45834 | RADLEX | RadLex, 4_1 |
| 198630 | RXNORM | RxNorm Vocabulary, 20AA_200908F |
| 349474 | SNOMEDCT_US | US Edition of SNOMED CT, 2020_09_01 |
| 167 | SOP | Source of Payment Typology, 9 |
| 4810 | SPN | Standard Product Nomenclature, 2003 |
| 222 | SRC | Metathesaurus Source Terminology Names |
| 28738 | UMD | UMDNS: product category thesaurus, 2020 |
| 4978 | USP | USP Compendial Nomenclature, 2020_02_06 |
| 1605 | USPMG | USP Medicare Model Guidelines, 2020 |
| 61112 | UWDA | University of Washington Digital Anatomist, 1.7.3 |
| 35585 | VANDF | Veterans Health Administration National Drug File, 2020_08_03 |
Overall Counts (May 2021)
| Concepts | Relationships | Attributes | Total Number of Terms |
|---|---|---|---|
| 3,795,128 | 24,431,718 | 27,255,262 | 8,300,055 |