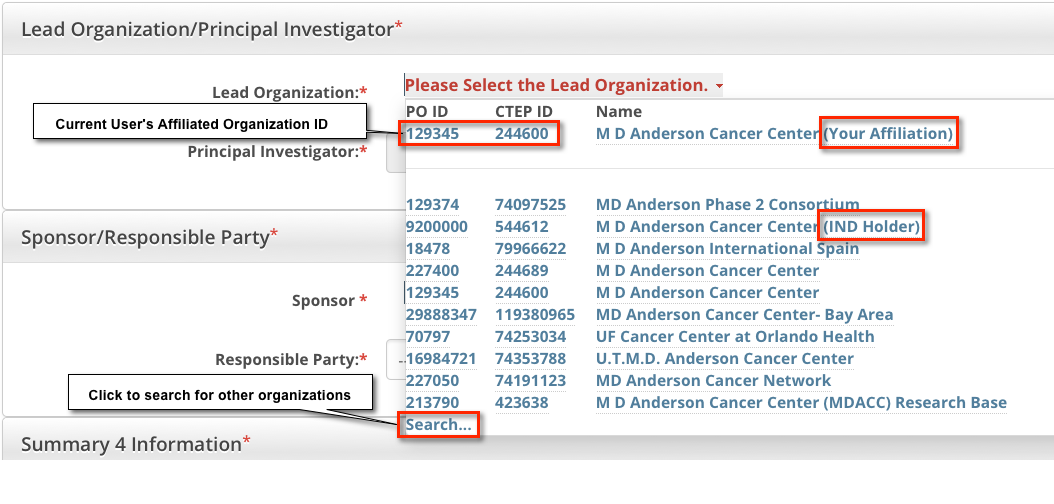
The Lead Organization is the institution that is the principal administrative organization responsible for the study. Each trial can have one Lead Organization only.
You must complete both fields in the Lead Organization/Principal Investigator section.
The Principal Investigator is the individual who is responsible and accountable for conducting the clinical trial. The PI assumes full responsibility for the treatment and evaluation of human subjects, and for the integrity of the research data and results. |
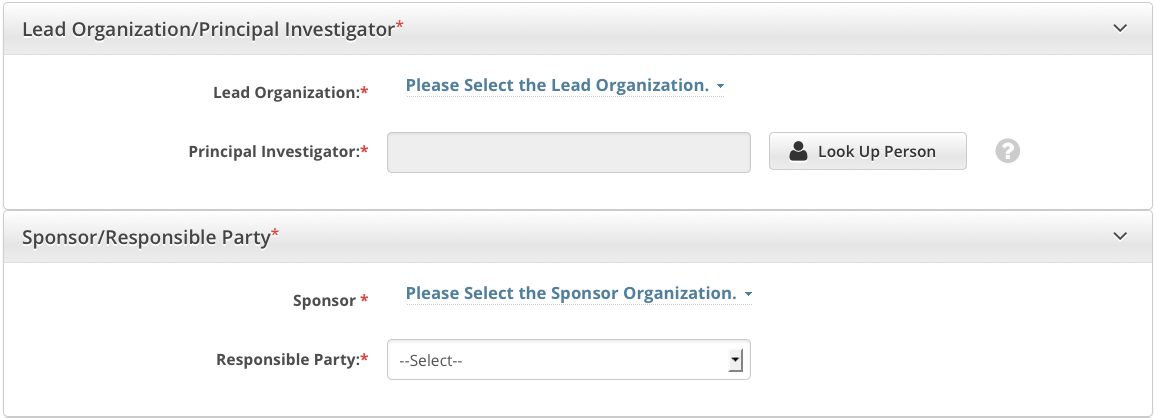
How to Complete the Lead Organization/Principal Investigator Section