
This section describes how to register new National, Externally Peer-Reviewed, and Institutional trials (collectively referred to as Complete trials) in the CTRP system, and how to import Industrial trials from ClinicalTrials.gov for registration in the CTRP. It applies to both interventional and non-interventional trials. The system logs you off if it detects that you have not used the application for 90 minutes. |

|
|
About Trial Registration
The way in which you register trials in the CTRP depends on a combination of the trial's Data Table 4 Categorization as either Complete or Abbreviated , your affiliated organization and its role in the trial, and whether the trial currently is registered in ClinicalTrials.gov with an NCT ID.

Guidelines for Complete Trials
If your trial is Complete, follow the instructions in Registering New Complete Trials.
- Cancer Center trials must meet the following criteria to be eligible for registration:
- Trials must have been active as of January 1, 2009 or any time thereafter.
- Submitting organization is the Lead Organization or the Coordinating Center.
- CTEP or DCP PIO-managed trials must meet the following criteria to be eligible for registration:
Guidelines for Abbreviated Trials

If your trial is Abbreviated, registration differs according to a combination of the following trial attributes:
- NCT ID (which indicates that the trial has been registered with ClinicalTrials.gov)
- Cancer Center type (NCI-designated Cancer Centers or other centers)
- NCI grant
- Lead organization
Guidelines for Trials with NCT IDs
If the trial is an Industry funded trial, and has an NCT ID, then it can be imported into CTRP from ClinicalTrials.gov in most cases. In some special cases, however, the trial cannot be imported directly and instead you must contact the CTRO for assistance.
- If your organization is not an NCI-designated Cancer Center organization, NIH institute, or pharmaceutical company, you will need to import the trial directly from ClinicalTrials.gov. Instructions for this process can be found under Registering Abbreviated (Industrial and Other) Trials.
- If your organization is the Lead Organization on a trial, and your organization is an NCI-designated Cancer Center organization, do not import the trial from ClinicalTrials.gov. Instead, submit the trial to CTRP as a Complete trial, as described in Registering New Complete Trials.
- If your organization is the Lead Organization for any trial on anything other than an NCI-designated Cancer Center organization, NIH institute, or pharmaceutical company trial, the CTRO contacts the Center and further categorizes the trial as Other/National or Other/Externally Peer-Reviewed based on whether your trial is conducted under an NIH grant, as follows:
- If your trial is conducted under an NCI grant, the CTRO categorizes it as a Consortia trial.
- If your trial is not conducted under an NCI grant, the CTRO categorizes it as follows:
- Other/National, for trials sponsored by an NIH institute
- Other/Externally Peer-Reviewed, for trials managed by a hospital or a center other than one designated by the NCI
Guideline for Trials without NCT IDs
If your trial is Abbreviated but does not have an NCT ID, please contact the CTRO for assistance at ncictro@mail.nih.gov. CTRO registers these trials manually.
Return to top of page
Registering New Complete Trials
Before you begin to register a trial, ensure that the trial does not exist in the system already by searching for trials using any of the criteria as per the instructions in Searching for Trials . The system uses the Lead Organization ID, Lead Organization Trial ID, and the ClinicalTrials.gov Identifier to detect duplicates. If a duplicate is detected, the system will not record your trial. |
How to Register New Complete Trials On the toolbar, click Register Trial, and select your trial's Submission Category (funding source) from the drop-down list, either National, Externally Peer-Reviewed, or Institutional.
The Register Trial page appears.  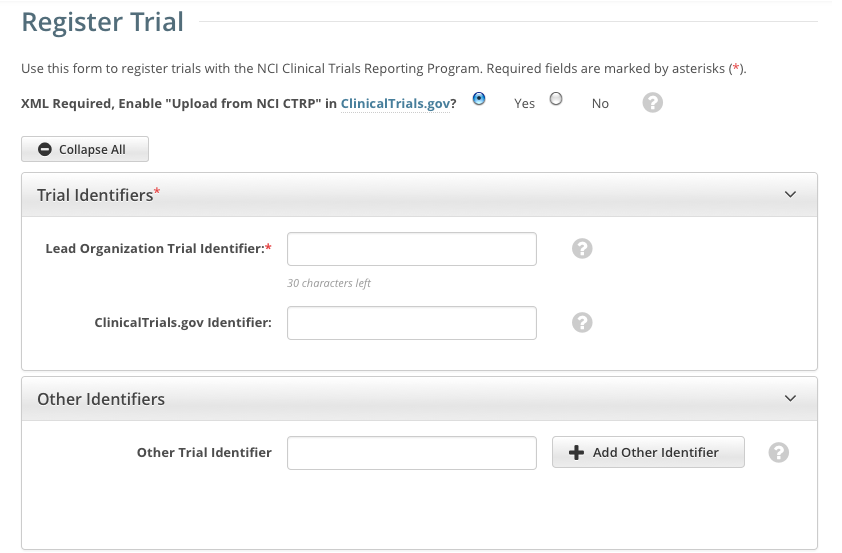
The system can create an XML document that is formatted to facilitate trial registration with ClinicalTrials.gov. The document it creates contains all the information that you submit during registration and all the trial data abstracted by the CTRO. If you indicate that you do not need to register the trial with ClinicalTrials.gov, you will not be asked to provide regulatory and responsible party information. By default, all sections of the registration form are displayed. To collapse or expand each section individually, click the Collapse or Expand icon on the right side of the section title as shown in the figures below. To collapse all sections, click Collapse All. |
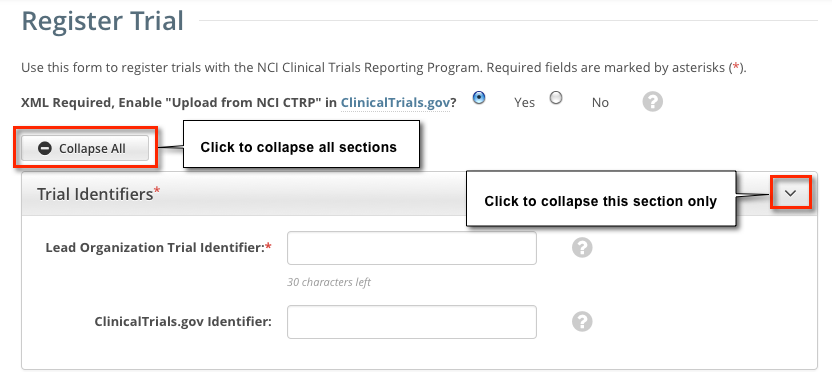
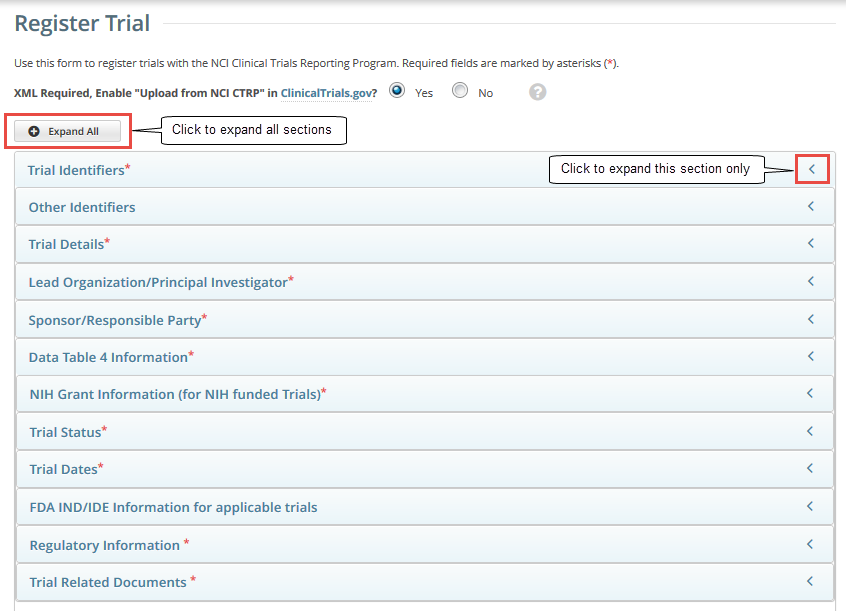
Select or enter the appropriate information in the text fields and drop-down lists. Fields are described in the following table. Be sure to provide information for all fields marked with an asterisk (*). If you cannot complete the registration of a trial in one Registration session, you can save a draft of the trial details you have completed. Later you can return to complete the registration in another session. |

Instructions for registering Complete trials XML required. Enable "Upload from NCI CTRP" in ClinicalTrials.gov? | If you require an XML document to register your trial with ClinicalTrials.gov, select Yes.
If you are not going to submit your trial to ClinicalTrials.gov, select No.
The option you select here dictates which sections you will be required to complete. For example, if you select No, you will not be required to complete responsible party and regulatory information. If you select Yes, NCI will be added as a collaborator to the Funding Source. | Various | Select or enter the appropriate information in the text fields and drop-down lists as appropriate according to the detailed instructions provided for each of the following sections: - Recording Trial Identification Information
- Recording Trial Details
- Recording Lead Organizations and Principal Investigators
- Recording Sponsors and Responsible Parties
- Recording Data Table 4 Information
- Recording NIH Grants
- Recording Trial Statuses
- Recording Trial Dates
- Recording INDs and IDEs
- Recording Regulatory Information
- Recording Trial-Related Documents
|  Save as Draft Save as Draft | Click to save a draft of the record so that you can complete the registration at another time. You must have provided, at the minimum, both the Lead Organization and Lead Organization Trial Identifier to save a draft.
The system saves your draft, assigns it a unique ID (for tracking purposes), and sends you an email message confirming that the information has been saved. You can end your Registration session and retrieve your draft later to complete the registration. | Review Trial | Click to initiate the system check for errors and missing information. The system displays the results in a message at the top of the Review Trial Details page. Indicators mark specific fields that you must complete or correct in order to submit the trial.
The Review Trial Details page is read-only. To make changes to the trial data, follow the instructions in Editing Trial Details. | | Cancel | Click to cancel the registration. A pop-up message prompts you to confirm cancellation. If you choose to cancel the registration, you will lose all data that you may have entered. |
|
Correct any errors if indicated, and repeat the previous steps as many times as necessary until the trial is error-free. To continue with the trial registration, scroll to the bottom of the Review Trial Details page, and then click Submit. To prevent creating a duplicate record, do not click Submit more than once. If you have to make changes after you click Submit, contact the CTRO at ncictro@mail.nih.gov rather than using your browser's Back button to make changes.
The registration notification message system sends you an email message to acknowledge that the trial has been submitted. Later it sends another email message to notify you when your trial has been accepted or rejected.
After submission, most users other than the trial submitter can not see the trial information you provided until the information has been validated. However, an organization administrator (if one exists) and an assigned owner can access the information prior to validation. |
|
Return to top of page
Recording Trial Identification Information

Return to top of page
Recording Trial Details


Return to top of page
Recording Lead Organizations and Principal Investigators

Return to top of page
Recording Sponsors and Responsible Parties

Return to top of page
Recording Data Table 4 Information

Return to top of page
Recording NIH Grants

Return to top of page
Recording Trial Statuses

Return to top of page
Recording Trial Dates

Return to top of page
Recording INDs and IDEs

Return to top of page
Recording Regulatory Information

Return to top of page
Recording Trial-Related Documents

Return to top of page
Converting Documents to PDFs

Return to top of page
Registering Abbreviated (Industrial and Other) Trials

You can register Industrial/Other trials in the CTRP by importing them directly from ClinicalTrials.gov. You must enter a ClinicalTrials.gov Identifier (NCT ID) for each trial you register. If the trial you want to register does not have an NCI ID, or if you do not know it, contact the CTRO for assistance at ncictro@mail.nih.gov.     The system registers the trials you import from ClinicalTrials.gov as Abbreviated trials. To classify a trial as "Other", contact the Clinical Trials Reporting Office staff at ncictro@mail.nih.gov after importing/registering the trial in the CTRP system.  For more information about Data Table 4 categorization, see Guidelines for Abbreviated Trials. |
How to Register Industrial Trials On the toolbar, click Register Trial, and select Industrial/Other. To read a definition of each of the trial submission categories (study sources), click View Trial Category Definitions, or, click the Help icon ( ) next to each category. ) next to each category. |

The Import ClinicalTrials.gov Trials page appears. 
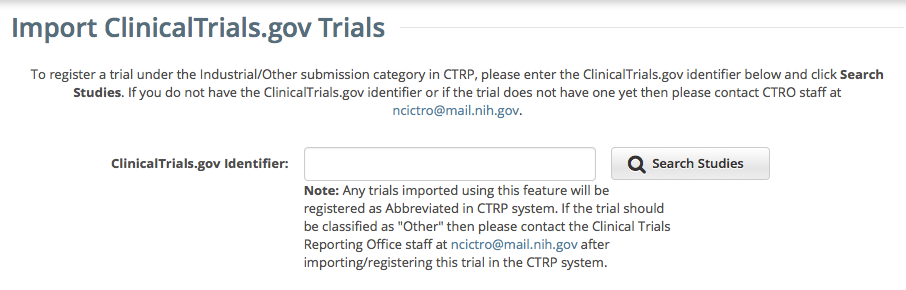 Enter the ClinicalTrials.gov Identifier, and then click Search Studies. The system searches for the ID you entered. If it finds a match in the CTRP, you can not import the trial. If the system does not find a match in the CTRP, the trial record from ClinicalTrials.gov appears.
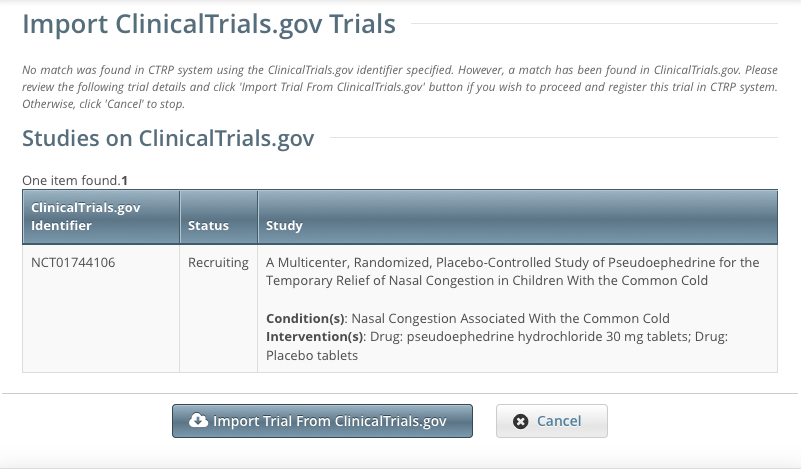 Click Import Trial From ClinicalTrials.gov. While it is possible for two users to attempt to import a trial at the exact same time, the system cannot process simultaneous imports. If you receive an error message the first time you attempt to import a trial, wait a short while, and then try again. |
The trial is registered in the CTRP and assigned a unique NCI identifier with the processing status Submitted. The system synchronizes the imported record in the CTRP with the one in ClinicalTrials.gov.  
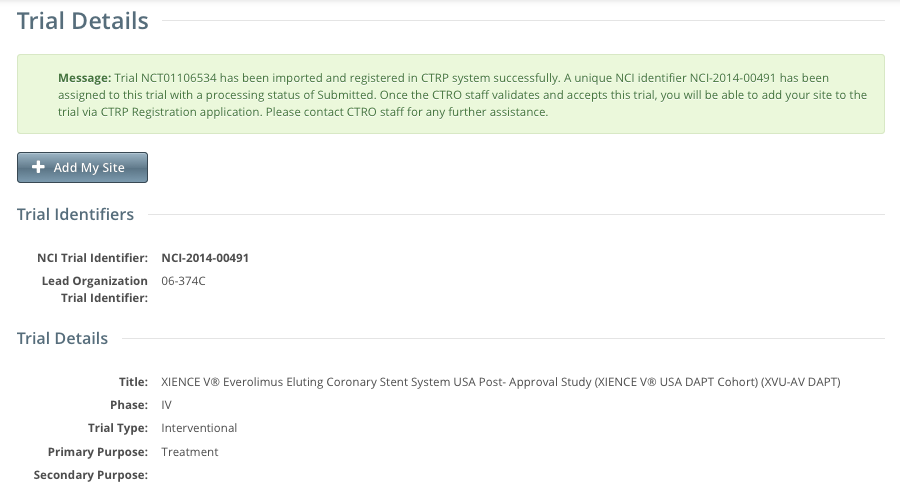 To add your site as a participant in the trial, click Add My Site. The Add Participating Site page appears. 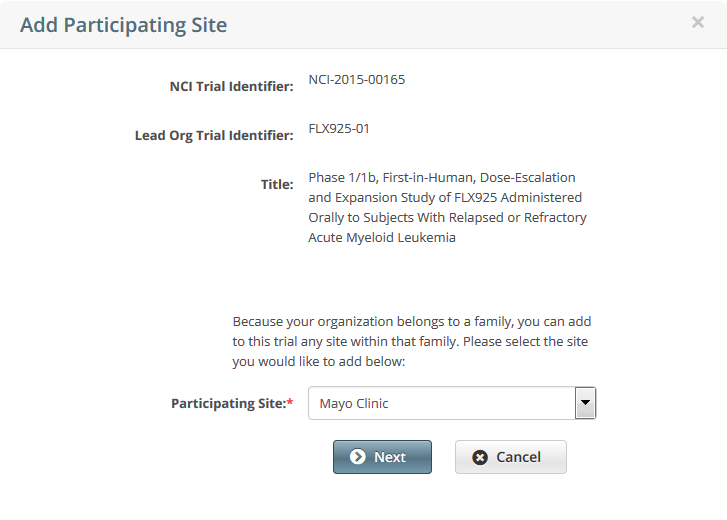
The Participating Site list contains all organizations in the Organization Family associated with your CTRP account.From the Participating Site list, select the organization that you want to add to this trial. Click Next. Another Add Participating Site page appears. 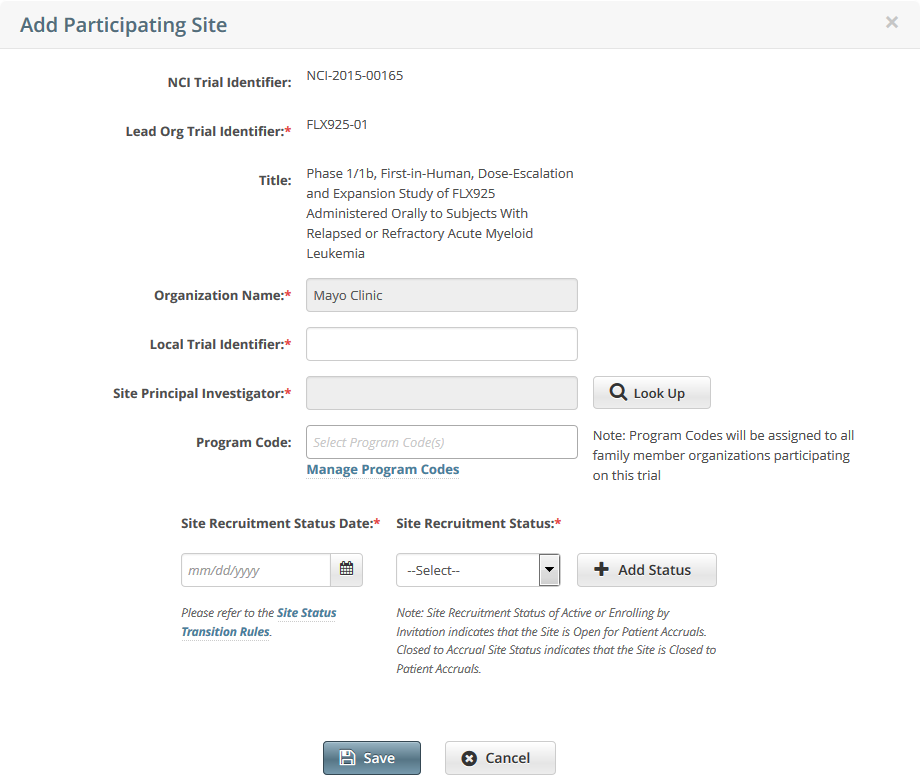 Complete the fields as per the instructions in Adding Your Site to Abbreviated Trials, and then click Save. The system sends you an email message when the CTRO has accepted the trial for registration in the CTRP. If your trial is not Industrial, contact the CTRO at ncictro@mail.nih.gov to request categorization of the trial as either National or Externally Peer-Reviewed.
The system synchronizes Industrial and Other trials currently registered in the CTRP with ClinicalTrials.gov trials every night by comparing their ClinicalTrials.gov Identifiers. The system updates CTRP trial records with the data imported from ClinicalTrials.gov if it finds matching records. 
The CTRP system does not import Person information from ClinicalTrials.gov. |
|
Return to top of page
Printing Trial Information

You can print a copy of the trial details to facilitate the review and/or keep for your records. You must review the trial in order to access the print feature. How to Print Trial Information - Scroll to the bottom of the Register Trial page, and then click Review Trial. If necessary, provide any missing information.
- Scroll to the bottom of the Review Trial Details page, and then click Print.
|
Return to top of page
Editing Trial Details

You can edit the details of the trial that you are registering before you submit the trial to the CTRP. If you want to edit a registered trial that you own, follow the instructions in Updating Trials. How to Edit Trial Details - Scroll to the bottom of the Register Trial page, and then click Review Trial.
- Scroll to the bottom of the Review Trial Details page, and then click Edit.
The Register Trial page displays all information you have provided in editable form. - Make changes as necessary, and then click Review Trial.
- After you have reviewed your edits, click Submit.
|
Return to top of page
Completing and Deleting Saved Drafts

During the course of registering a trial, you can save the information you have already provided as a draft , and return to it later. Once you retrieve your draft, you can complete the registration or delete the draft altogether. How to Complete or Delete Saved Drafts - Retrieve the draft by searching for it using the Search Saved Drafts option. (For details, see Searching for Trials .)
Saved draft records are listed on the Search Results page.
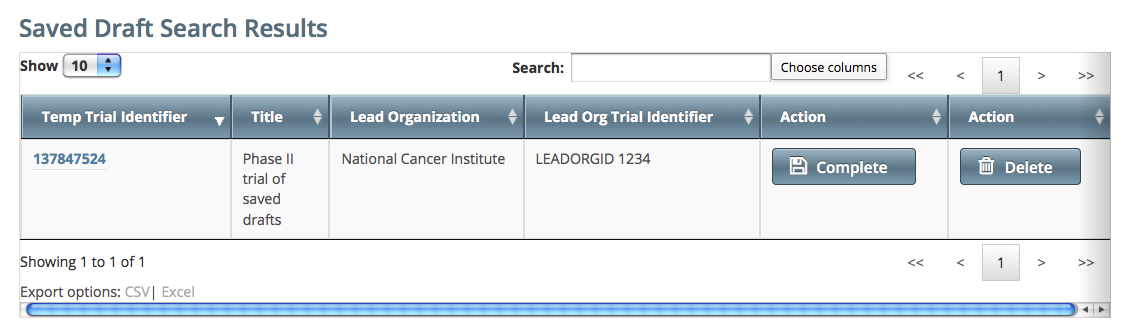 - Do one of the following in the appropriate Action column on the right side of the table:
- To delete the record, click Delete. The draft record is deleted from the system permanently.
- or - - To complete the record, click Complete.
The Register Trial page displays all the information you provided previously. You can complete the rest of the information by following the steps in Registering a Complete Trial. |
Return to top of page