Search for the trial of interest. For instructions, refer to Searching for Trials in PA.
In the search results, Select the NCI Trial Identifier link for that trial. The Trial Identification page opens.
On the Trial Identification page, check out the trial. For instructions, refer to Checking In and Checking Out Trials. (This checkout step is optional for Super Abstractors.)
On the Administrative Data menu, click NCI Specific Information. The NCI Specific Information page opens. 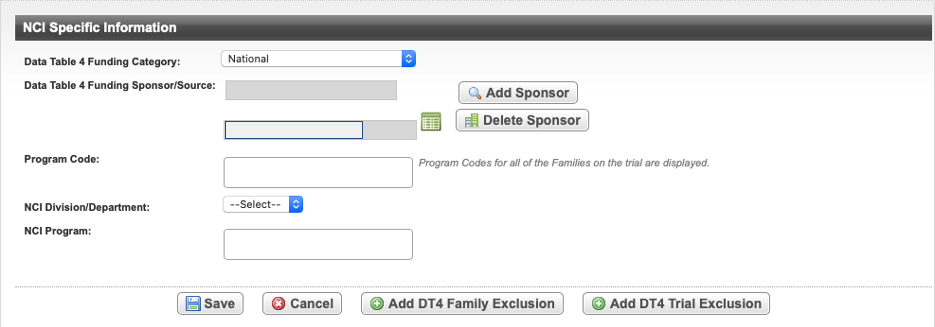
The following table describes the fields on the NCI Specific Information page.
Field Label | Description/Instructions | |
|---|---|---|
Data Table 4 Funding Category | Select a trial type based on the role/responsibility/participation in the study. For information, refer to CTRP Trial Categories, Study Sources and https://cancercenters.cancer.gov/GrantsFunding/eData#dt4. | |
Data Table 4 Funding Sponsor/Source | To add a sponsor, select Add Sponsor and search for the name of the external sponsor or funding source as defined by the Data Table 4 report. (See Searching for Organizations.)
To delete an existing sponsor, select Delete Sponsor. Refer to https://cancercenters.cancer.gov/GrantsFunding/eData#dt4 for further information about specific Funding Sponsors. | |
| Industrial? | For Abbreviated trials, indicate whether the trial is an Industrial trial, or other category, according to the matrix in Industrial Values. | |
Program Code | Only available if the Lead Organization for the trial belongs to an organization family. The Program Code field lists all program codes available for the organization family of the lead organization. Select one or more program codes. The program codes are generally entered by the trial submitter. To view or modify a different family's program codes, refer to the Registration Site Administration chapter of the Registration User's Guide. | |
| Send Trial Information to ClinicalTrials.gov? | For Complete, NCI Sponsored trials, select one of the following to indicate whether to include this trial in the nightly batch upload to ClinicalTrials.gov:
For further information about this field, see Conditions for Sending Trial Information to ClinicalTrials.gov. | |
| Comments | Enter a comment about your selection. | |
| NCI Division/Department | Select the division or department that is managing the trial. | |
| NCI Program | Select one or more relevant programs. |