This page provides an overview of the changes in CTRP. For any questions or issues regarding these feature enhancements, please contact the CTRO (NCICTRO@mail.nih.gov).
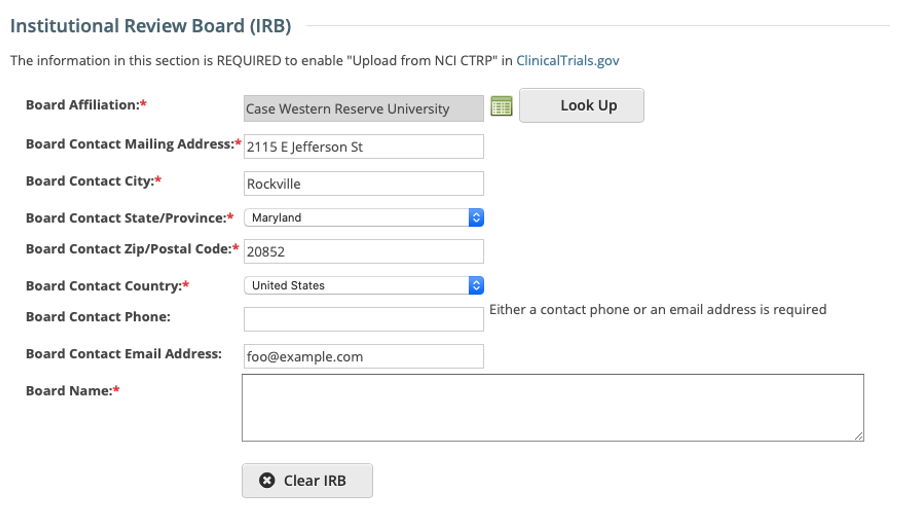
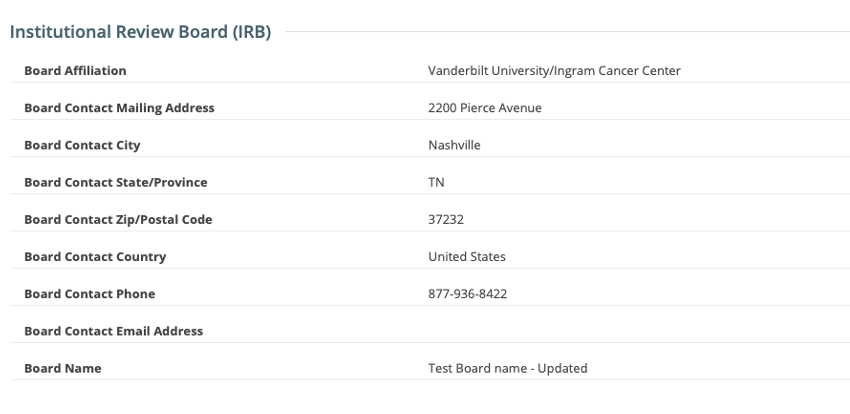
IRB fields are now exposed in the CTRP Registration application. IRB information can be added/modified when registering, updating/verifying and amending non-industrial trials. IRB fields are read-only when viewing trials on the Trial Details Page.
Registering, Amending or Updating/Verifying a non-industrial trial: IRB fields are modifiable

Viewing a trial on the Trial Search page: IRB fields are read-only
For additional information about the IRB fields in the CTRP Registration application, please visit these CTRP User Guide pages:
Recording Institutional Review Board (IRB) Information
Updating Institutional Review Board (IRB) Information in Complete Trials
Transition to the NCIs multi-factor authentication system, Okta: Your CTRP username is now your institutional email address.
Migration of CTRP user accounts to Okta. Okta multi-factor authentication is used to provide additional security beyond just entering a username and password. Each Okta account requires the configuration of at least one of the following methods:
For additional information about creating and setting up an Okta account, please visit these CTRP User Guide pages:
Updates to the Data Table 4 (DT4) Anatomic Site values displayed on the CTRP-generated DT4 report:
Previous DT4 Anatomic Site Value | Updated DT4 Anatomic Site Value |
Leukemia, not otherwise specified | Leukemia, other |
Breast – Female | Breast |
Breast – Male | Breast |
Hodgkin's Lymphoma | Hodgkin Lymphoma |
Non-Hodgkin's Lymphoma | Non-Hodgkin Lymphoma |
Soft Tissue / Sarcoma | Soft Tissue |
Note: For trials with numerous “Primary Sites” (DT4 Anatomic Sites), the value “Multiple” for the DT4 Anatomic Site Code will be used and displayed on the CTRP-generated DT4 report. All existing trials in CTRP have been updated to reflect this change as well.
For a complete list of DT4 anatomic site values displayed on the CTRP-generated DT4 report, please visit this CTRP User Guide page:
https://wiki.nci.nih.gov/display/CTRPdoc/Data+Table+4+Anatomic+Site+Values
Removal of the following fields from the Trial Summary Report (TSR):
CTRP Trial Record Verification: Improved the user experience for CTRP trial owners, trial submitters and site administrators with Verifying, Updating and/or Amending their trial records. Key highlights from this enhancement include:
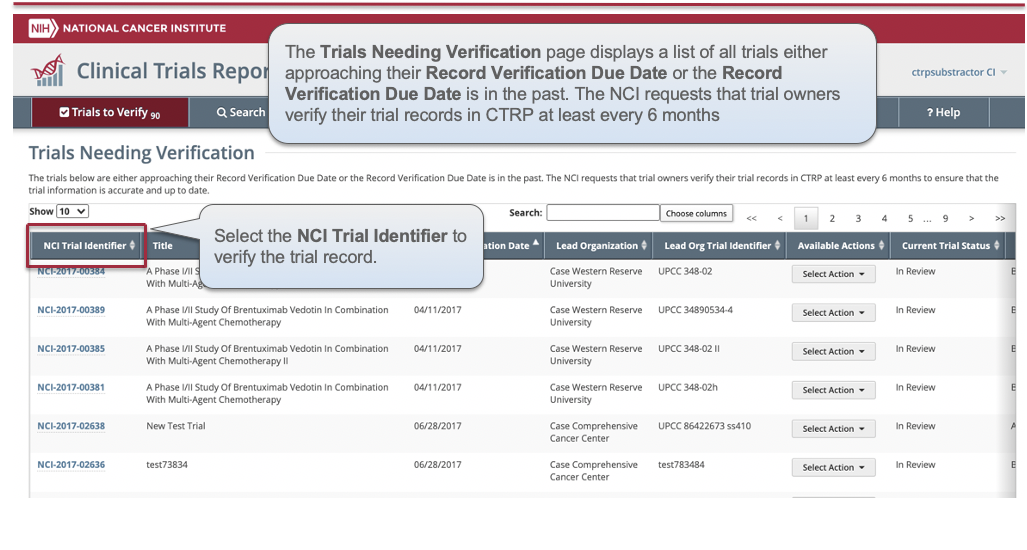
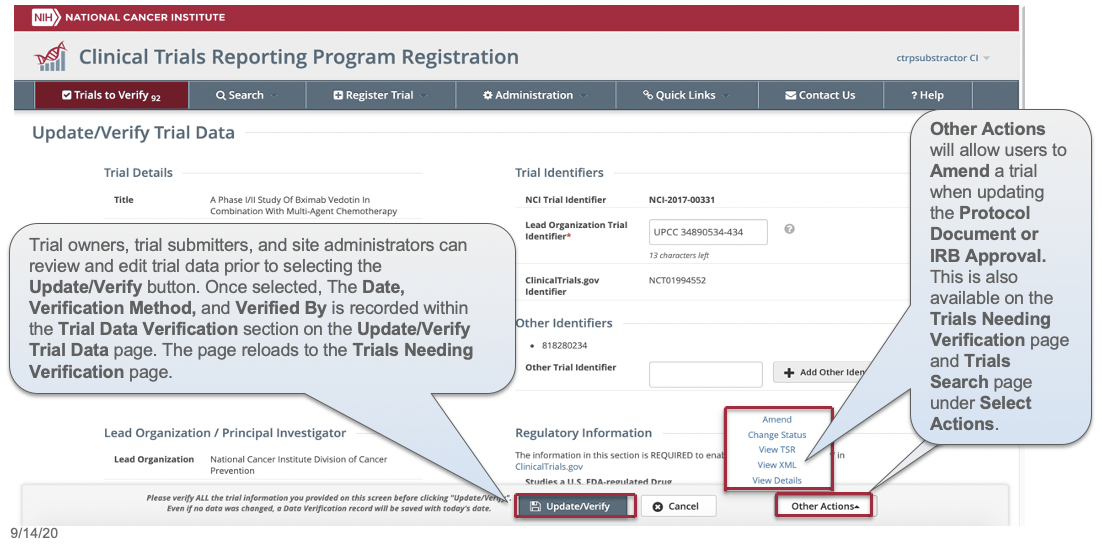

For more information on the CTRP Trial Record Verification processes, please visit these CTRP User Guide pages:
https://wiki.nci.nih.gov/display/CTRPdoc/Updating+Trial+Information
https://wiki.nci.nih.gov/display/CTRPdoc/How+to+Verify+Trial+Data
https://wiki.nci.nih.gov/display/CTRPdoc/How+to+Access+the+Trial+Data+Verification+Page
https://wiki.nci.nih.gov/display/CTRPdoc/Amending+Trials
Flexible Accrual: Enhanced the CTRP accrual reporting capabilities to include more “Flexible Accrual” reporting options. Centers can now request to update the default accrual reporting type on a study registered in CTRP. Changing the default accrual reporting type requires a ticket to be submitted to the CTRO (NCICTRO@mail.nih.gov) for review and approval, before any change is granted.
The current default accrual reporting types are as follows:
Trial Type | Default Accrual Reporting Type |
Complete Interventional | Subject |
Complete Non-Interventional | Subject |
Abbreviated/Imported Interventional | Summary |
Abbreviated/Imported Non-Interventional | Summary |
For more information on the CTRP Accrual process, including Flexible Accrual, Partial Subject accrual and other changes made within the Accrual application (e.g., ability to Delete All Trial Study Subjects,) please visit these CTRP User Guide pages:
https://wiki.nci.nih.gov/display/CTRPdoc/About+Accrual
https://wiki.nci.nih.gov/display/CTRPdoc/Searching+for+Trials+in+Accrual
If you would like to receive future CTRP announcements you may add yourself to the list here: https://list.nih.gov/cgi-bin/wa.exe?SUBED1=ctrp-users-l&A=1