You can add one or more study subject accrual records for any study to which you have been granted access.
1 If necessary, refer to Requesting Permission to Submit Accrual Data.
Study subject records include demographic data as well as the disease name.
How to Add Study Subject Records
- Select the study you want to work with by following instructions in Searching for and Selecting Your Trials, and clicking the corresponding NCI Trial Identifier link. The Search Study Subject page appears.
- Click Add New Study Subject.
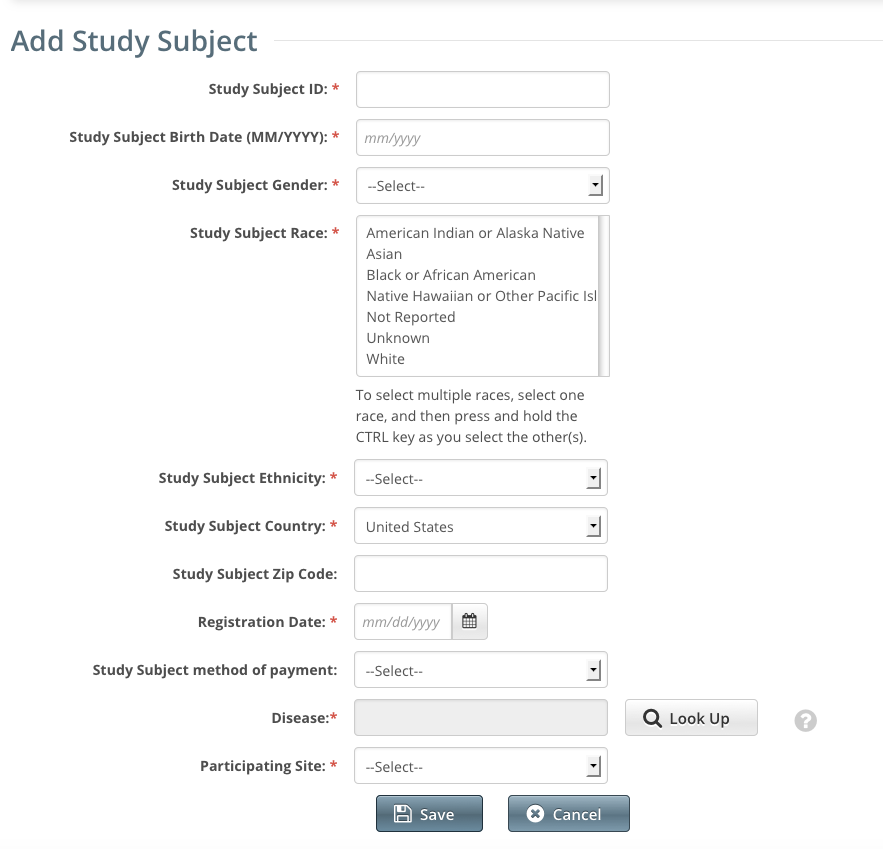
The Add Study Subject page appears. (Screenshot below displays Subject accrual)
Enter the required demographic information in the text fields and drop-down lists
.An asterisk (*) indicates a required field.The following table describes the valid values and formats for each field.
Accrual Data Elements for Complete Studies
Descriptions and instructions for Study Subject and Partial Subject demographic data fieldsRequired/Conditional Accrual Data Element Definition Subject Partial Subject Accepted Values Study Subject ID Enter the unique Patient ID as per the lead organization or the study site where the subject is registered. R R Any numeric or alphanumeric value Study Subject Birth Date (MM/YYYY) Enter the subject's month and year of birth in the format MM/YYYY.R Accrual application format: MM/YYYY
REST Services format: YYYY-MM-DD
Batch Upload format: YYYYMMStudy Subject Gender Select the subject’s biological sex. If biological sex information is not available, select Unknown. R Male
Female
Unspecified
Undifferentiated
UnknownStudy Subject Race Select one or more values for race.
To select multiple races, select one race, and then press and hold the CTRL/CMD key as you select the other(s).
R American Indian or Alaska Native
Asian
Black or African American
Native Hawaiian or Other Pacific Islander
Not Reported
Unknown
WhiteStudy Subject Ethnicity Select a value for ethnicity. R Hispanic or Latino
Not Hispanic or Latino
Not Reported
UnknownStudy Subject Country Select the study subject's country of residence. R Study Subject Zip Code Enter the study subject’s zip code. C C Required if the Study Subject Country is the US, US territories and outlying islands.
Must be in a 5 digit or 9 digit (DDDDD-DDDD) format
Registration Date Enter the date that the subject was registered on the study. R R Accrual application format: MM/DD/YYYY
REST Services format: YYYY-MM-DD
Batch Upload format: YYYYMMDDDisease
Click Look Up, and follow the instructions in Selecting Diseases for Study Subject Records.
Note: For ICD-O-3, CTRP uses a hybrid model with the codes from IACR and the codes in NCI SEER that are not available in IACR.
R Partial Subject:
Disease is not required.
ICD-O-3: If Disease is reported, Site is also required.
Study Subject:Disease Code is required for all trial types.
ICD-O-3: Disease and Site are required.
Site
(for ICD-O-3 Disease Codes)
Click Look Up, and follow the instructions in Selecting Sites for Study Subject Records Using ICD-O-3 Codes.
Site Code information is available at:
http://www.iacr.com.fr/index.php?option=com_content&view=category&layout=blog&id=100&Itemid=577
Note: CTRP uses a hybrid model with the codes from IACR and the codes in NCI SEER that are not available in IACR.
C Partial Subject:
Site is not required.
ICD-O-3: If Site is reported, Disease is also required.
Study Subject:Site is required for all trial types.
ICD-O-3: Site and Disease are required.
Participating Site Select the appropriate site from the drop-down list. R R Study Subject method of payment Select the appropriate payment method. Private Insurance
Medicare
Medicare and Private Insurance
Medicaid
Medicaid and Medicare
Military or Veterans Sponsored, NOS
Military Sponsored (Including CHAMPUS & TRICARE)
Veterans Sponsored
Self-Pay (No Insurance)
No Means of Payment (No Insurance)
Managed Care
State Supplemental Health Insurance
Other
Unknown- Click Save.
The study subject record appears in the List of Study Subjects.
For Complete studies, lead organizations report all subjects accrued for the study (both in the lead organization and in all participating sites). For Abbreviated studies, each participating site reports the number of its own accruals (accrual count) only.
---
- If your organization currently reports subject accrual information for studies to CTEP or DCP via the CDUS/OPEN systems, continue to report subject accrual information via CDUS/OPEN. The NCI will manage the transfer of subject accrual data for CDUS/OPEN studies internally. Otherwise, report your organization's subject accrual data to CTRP.