Group/cohort information pertains to Complete non-interventional trials only. In non-interventional studies, cohorts are groups of individuals, initially defined and composed, with common characteristics (e.g., condition, birth year), who are examined or traced over a given time period.
When adding groups to a trial, you assign to them the interventions currently abstracted for the trial. For instructions on abstracting interventions, refer to Abstracting Interventions.
The following rules apply to each trial:
There must be at least one group/cohort that implies that the entire study subject population receives the same intervention.
The number of groups/cohorts added to the trial must coincide with the number indicated in the trial’s Design Details. See Abstracting Non-Interventional Trial Design Details.
- Every group/cohort must have at least one assigned intervention.
- Every intervention must be assigned to at least one group/cohort.
- A trial must have only one group with the same combination of description and associated intervention.
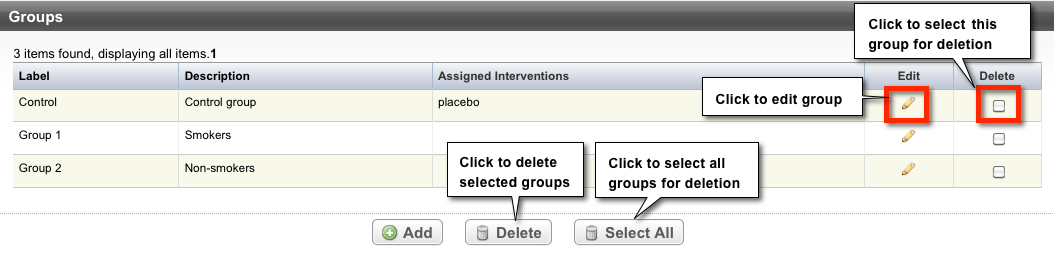
Trials can have multiple group/cohort records. Each record displays the following group information:
| Field | Definition |
|---|---|
Label | Short name used to identify the group |
Description | Explanation of the nature of the study group (e.g., those with a condition and those without a condition; those with an exposure and those without an exposure). |
Assigned Interventions | Intervention assigned to a given group |
Group Records
For instructions, refer to the following pages: