You must complete both fields in the Lead Organization/Principal Investigator section.
Tip
The Principal Investigator is the individual who is responsible and accountable for conducting the clinical trial. The PI assumes full responsibility for the treatment and evaluation of human subjects, and for the integrity of the research data and results.
How to Complete the Lead Organization/Principal Investigator Section
- Next to the Lead Organization field, click Please Select the Lead Organization.
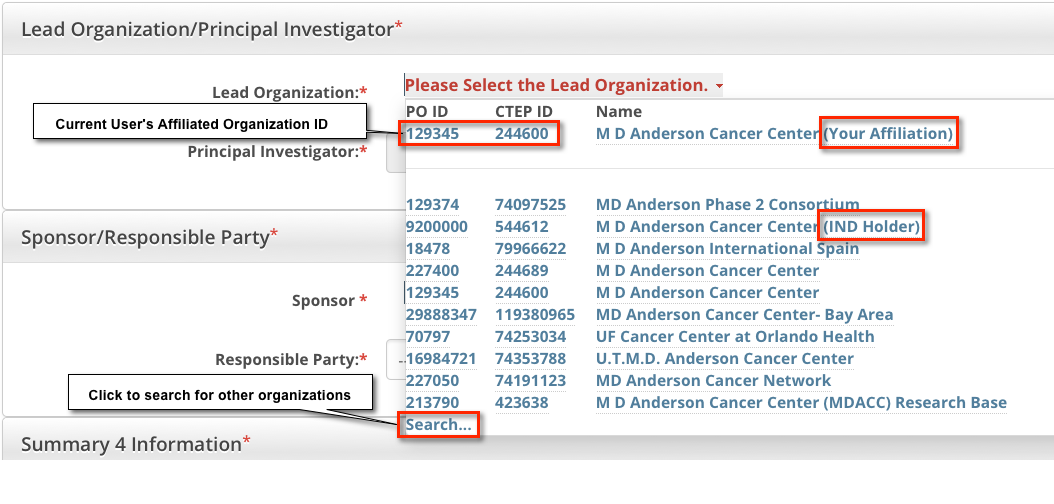
The system displays your affiliated organization first, followed by affiliated organization family members. Organizational roles are displayed when applicable. - Select the appropriate organization from the list. If the organization is not listed, click Search, and search for the organization as per the instructions in Looking up Registered Organizations. (If the search does not return your trial's lead organization, you can ask the CTRO to create the organization record. To do so, follow the instructions in Requesting Creation of an Organization Record.)
- Next to Principal Investigator click Look Up Person, and search for the principal investigator as per the instructions in Looking Up Registered Persons. (If your trial's principal investigator's name is not listed, you can ask the CTRO to create the person record. To do so, follow the instructions in Requesting Creation of a Person Record.)