|
Page History
This page provides an overview of the changes in CTRP. For any questions or issues regarding these feature enhancements, please contact the CTRO (NCICTRO@mail.nih.gov).
July 2021
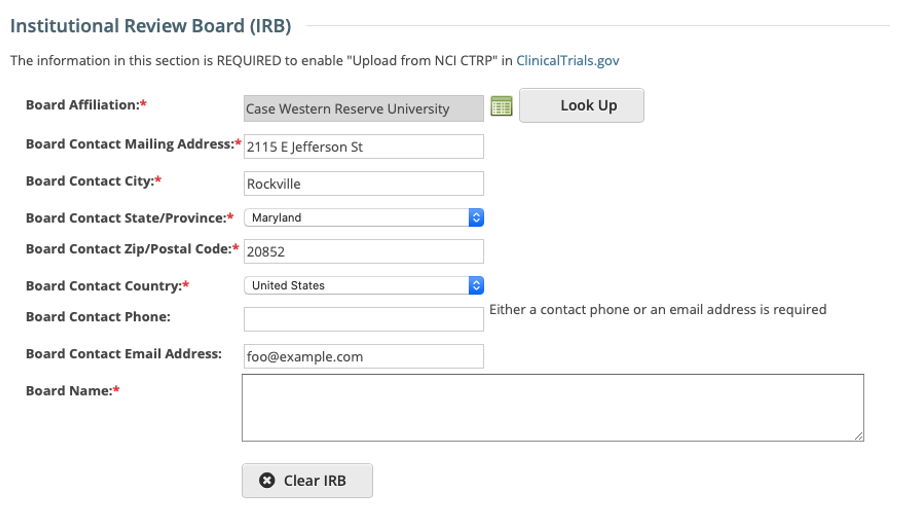
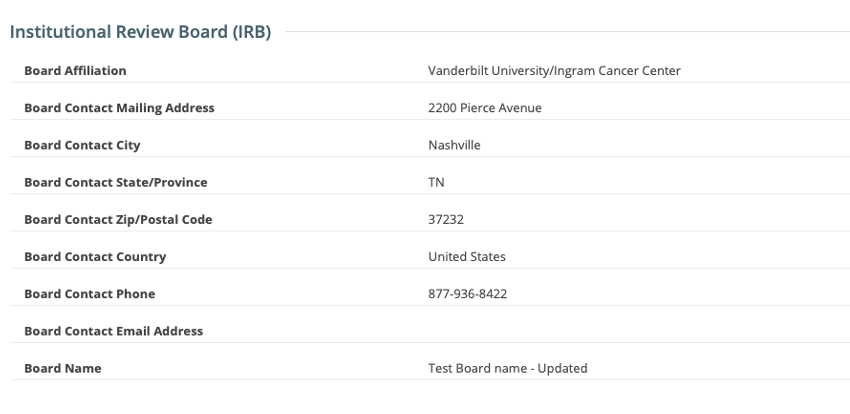
IRB fields are now exposed in the CTRP Registration application. IRB information can be added/modified when registering, updating/verifying and amending non-industrial trials. IRB fields are read-only when viewing trials on the Trial Details Page.
Registering, Amending or Updating/Verifying a non-industrial trial: IRB fields are modifiable
Viewing a trial on the Trial Search page: IRB fields are read-only
For additional information about the IRB fields in the CTRP Registration application, please visit these CTRP User Guide pages:
Recording Institutional Review Board (IRB) Information
Updating Institutional Review Board (IRB) Information in Complete Trials
June 2021
Transition to the NCIs multi-factor authentication system, Okta: Your CTRP username is now your institutional email address.
...