|
Page History
...
If the Trial Search page is not already displayed, on the Protocol Abstraction main menu on the left side of the page, click Trial Search. The Trial Search page appears.
HTML Comment hidden true Screenshot TBD.
Type or select any of the search criteria listed in the following table as appropriate.
Info You can select multiple values for Principal Investigator, Trial Phase, Processing Status, Search by Submission Type, Search by Submission Method, Lead Organization, Primary Purpose, Current Trial Status, and Milestone.
Search CriteriaField
Instructions/Description/Instructions
Official Title
Specify To narrow the search by this trial attribute, specify one or more words from the
Use wildcards as appropriate.Include Page Trial Attribute Official Title - Include v4.4 Trial Attribute Official Title - Include v4.4 Include Page Tip for searching by title - Include v4.4 Tip for searching by title - Include v4.4 For information, refer to Abstracting Trial Descriptions, Titles, and Identifiers.
Identifier Type
Specify the To narrow the search by both identifier and type of trial identifier, specify both. For information, refer to the identifier type descriptions in Abstracting Trial Descriptions, Titles, and Identifiers.
The Identifier Type field is optional, even when specifying an identifier.
Identifier
Specify To narrow the search by this trial attribute, specify all or part of the numeric or alphanumeric identifier assigned to the trial. For Inter-Group trials, specify the Lead Group's trial number. Consider the following examples:
- NCI-2000-00015
- ECOG-1234
For information, refer to Abstracting Trial Descriptions, Titles, and Identifiers.
Principal Investigator
Specify To narrow the search by this trial attribute, specify the
Include Page Trial Attribute PI - Include v4.4 Trial Attribute PI - Include v4.4 Tip title Search tip To narrow the list of investigators displayed, click the Principal Investigator field and type the initial letter of the last name. You can also use the Up and Down arrow keys on your keyboard to scroll through the list.
For information, refer to Abstracting Sponsors and Responsible Parties.
Trial Phase
Specify To narrow the search by this trial attribute, specify the
For information, refer to Trial Phase Value Definitions or Abstracting Interventional Trial Design.Include Page Trial Attribute Phase - Include v4.4 Trial Attribute Phase - Include v4.4 Processing Status
Specify To narrow the search by this trial attribute, specify the
For processing status definitions, refer to Trial Processing Statuses.Include Page Trial Attribute Current Trial Processing Status - Include v4.4 Trial Attribute Current Trial Processing Status - Include v4.4 Search by On-Hold StatusSelect the
On-Hold Status from the drop-down listTo narrow the search by this trial attribute, specify the hold status. Otherwise, select All. Valid values are as follows:
- All. Search for trials without consideration of On Hold status.
- On Hold. Limits your search to trials that are currently on hold.
- Not On Hold. Limits your search to trials that are currently not on hold.
Search by Submission Type
To narrow the search by this trial attribute, specify the
Otherwise, select All. Valid values are as follows:Include Page Trial Attribute Submission Type OUA - Include v4.4 Trial Attribute Submission Type OUA - Include v4.4 - All. Search for original and amended submissions
- Original. Search for original submissions only (indicated by “O” in the results list)
- Update. Search for updated submissions only (indicated by “U” in the results list)
- Amendment. Search for amended submissions only (indicated by “A” in the results list)
Trial Type
Specify To narrow the search by this trial attribute, specify the
Otherwise, select All. For information, refer to Trial Types and Subtypes, Abstracting Interventional Trial Design, or Abstracting Non-Interventional Trial Design.Include Page Trial Attribute Trial Type - Include v4.4 Trial Attribute Trial Type - Include v4.4 Trial Sub-type Specify To narrow the search by this trial attribute, specify the
For information, refer to Trial Types and Subtypes or Abstracting Non-Interventional Trial Design.Include Page Trial Attribute Trial Subtype - Include v4.4 Trial Attribute Trial Subtype - Include v4.4 Search by Submission Method
Select To narrow the search by this trial attribute, specify the means by which the trial was submitted to the CTRP from the drop-down list. Valid values are as follows: - Batch. Search for trials submitted via the NCI CTRP Registration Batch Upload site
- Registration. Search for trials submitted via Registration
- ClinicalTrials.gov. Search for trials imported from ClinicalTrials.gov
- PDQ. Search for trials submitted via the NCI Physician Data Query database
- Grid Services. Search for trials submitted via the NCI CTRP web services
- Other. Search for trials submitted via any method other than those listed above
Lead Organization
Specify To narrow the search by this trial attribute, specify the
Include Page Trial Attribute Lead Org Name - Include v4.4 Trial Attribute Lead Org Name - Include v4.4 Tip title Tip To narrow the list of organizations displayed, click the Lead Organization field and type the initial letter of the organization’s name. You can also use the Up and Down arrow keys on your keyboard to scroll through the list. For information, refer to Abstracting Sponsors and Responsible Parties.
Primary Purpose
Specify To narrow the search by this trial attribute, specify the
ForInclude Page Trial Attribute Primary Purpose - Include v4.4 Trial Attribute Primary Purpose - Include v4.4 For information, refer to Primary Purpose Value Definitions, Abstracting Interventional Trial Design, or Abstracting Non-Interventional Trial Design.
Current Trial StatusSpecify
trial status definitionsTo narrow the search by this trial attribute, specify the
ForInclude Page Trial Attribute Current Trial Status - Include v4.4 Trial Attribute Current Trial Status - Include v4.4 information, refer to Trial Status Values in the CTRP and ClinicalTrials.gov, Expanded Access Statuses, and Abstracting Trial Statuses.
Milestone
Specify To narrow the search by this trial attribute, specify the
For individual milestone definitions, see information, refer to Processing Trial Milestones or Milestone Definitions and Recording Rules. When appropriate, both Administrative and Scientific milestones and their dates are included for each trial in the search results list.Include Page Trial Attribute Current Milestone - Include v4.4 Trial Attribute Current Milestone - Include v4.4 Trials I Have Checked Out
Select this check box if you want to limit your To narrow the search to trials that you have checked out to validate, abstract, or edit, select this check box. For information, refer to Checking In and Checking Out Trials.
Search By Trial Category
To narrow the search by this trial attribute, specify the
For information about these categories, refer to CTRP Trial Categories (, Study Sources). Otherwise, select All.Include Page Trial Attribute Study Source Category - Include v4.4 Trial Attribute Study Source Category - Include v4.4 Search CTEP, DCP, or All Trials
Select To narrow the search by this trial attribute, specify the sponsor organization from the drop-down list. Otherwise, select All Trials. Valid values are as follows:
All Trials. Returns all trials, including CTEP and DCP trials
CTEP and DCP PIO. Returns either CTEP or DCP PIO trials
- CTEP PIO Trials Only. Returns CTEP PIO trials only
- DCP PIO Trials Only. Returns DCP PIO trials only
DCP is an abbreviation of Division of Cancer Prevention. CTEP is an abbreviation of Cancer Therapy Evaluation Program. PIO is an abbreviation of Protocol and Information Office. For information, refer to Abstracting Sponsors and Responsible Parties.
Click Search, or press the Enter key on your keyboard.
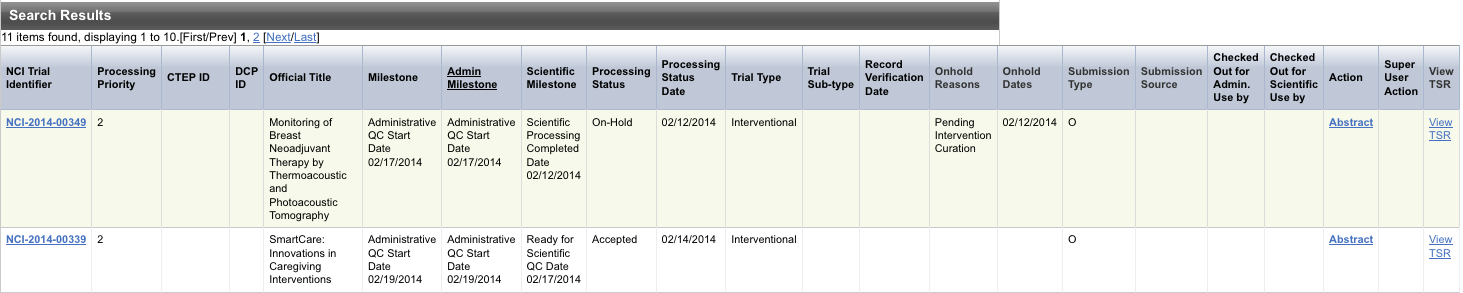
The results of your query are listed in the Search Results section.Anchor searchresults searchresults
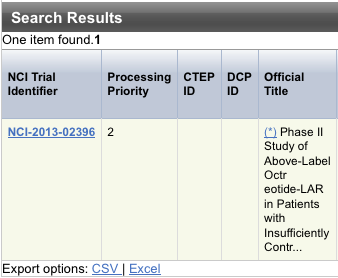
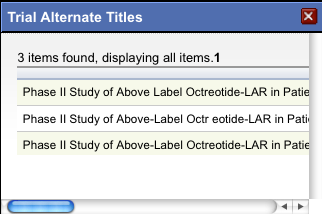
The system displays an asterisk (*) in the Official Title column for trials that have alternate titles.
To view the alternate titles, click the asterisk.
Alternate titles are listed in the Trial Alternate Titles window.
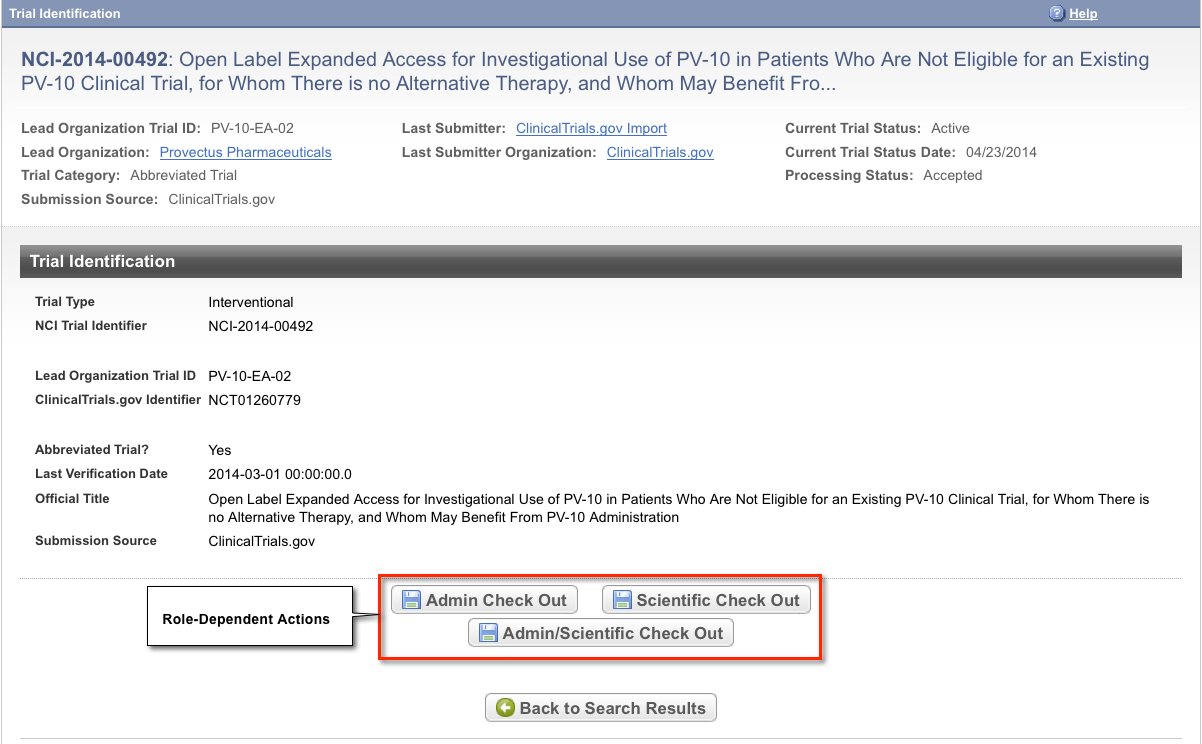
To view details for a trial, click the NCI Trial Identifier link for the trial of interest, or click any link in the Action column.
The Trial Identification page displays trial identifiers and other details. It also displays one or more options to check out the trial for processing. For example, if you logged in to the application as a Super Abstractor, you can check the trial out for both administrative and scientific processing, as in the figure below. However, if you logged in as an Administrative Abstractor, your only option is to check in/out the trial for administrative processing.
Tip title Returning to the Search Results page To return to your most recent search results, click Back to Search Results on the Trial Identification page. - To view a Trial Summary Report (TSR), click the View TSR link for the trial of interest, and open the file.
- To export the search result records, at the upper or lower right corner of the search results table, click one of the following:
- Click CSV to create a generic comma-separated value file.
- Click Excel to create a Microsoft Excel spreadsheet.
...