If the Trial Search page is not already displayed, on the Protocol Abstraction main menu on the left side of the page, click Trial Search. The Trial Search page appears.
Screenshot TBD. |
Type or select any of the search criteria listed in the following table as appropriate.
You can select multiple values for Principal Investigator, Trial Phase, Processing Status, Search by Submission Type, Search by Submission Method, Lead Organization, Primary Purpose, Current Trial Status, and Milestone. |
Field | Instructions/Description | |
|---|---|---|
Official Title | To narrow the search by this trial attribute, specify one or more words from the For information, refer to Abstracting Trial Descriptions, Titles, and Identifiers. | |
Identifier Type | To narrow the search by both identifier and type of trial identifier, specify both. For information, refer to the identifier type descriptions in Abstracting Trial Descriptions, Titles, and Identifiers. The Identifier Type field is optional, even when specifying an identifier. | |
Identifier | To narrow the search by this trial attribute, specify all or part of the numeric or alphanumeric identifier assigned to the trial. For Inter-Group trials, specify the Lead Group's trial number. Consider the following examples:
For information, refer to Abstracting Trial Descriptions, Titles, and Identifiers. | |
Principal Investigator | To narrow the search by this trial attribute, specify the
For information, refer to Abstracting Sponsors and Responsible Parties. | |
Trial Phase | To narrow the search by this trial attribute, specify the | |
Processing Status | To narrow the search by this trial attribute, specify the | |
Search by On-Hold Status | To narrow the search by this trial attribute, specify the hold status. Otherwise, select All. Valid values are as follows:
| |
Search by Submission Type | To narrow the search by this trial attribute, specify the
| |
Trial Type | To narrow the search by this trial attribute, specify the | |
| Trial Sub-type | To narrow the search by this trial attribute, specify the | |
Search by Submission Method | To narrow the search by this trial attribute, specify the means by which the trial was submitted to the CTRP. Valid values are as follows:
| |
Lead Organization | To narrow the search by this trial attribute, specify the
For information, refer to Abstracting Sponsors and Responsible Parties. | |
Primary Purpose | To narrow the search by this trial attribute, specify the | |
Current Trial Status | To narrow the search by this trial attribute, specify the | |
Milestone | To narrow the search by this trial attribute, specify the | |
Trials I Have Checked Out | To narrow the search to trials that you have checked out to validate, abstract, or edit, select this check box. For information, refer to Checking In and Checking Out Trials. | |
Search By Trial Category | To narrow the search by this trial attribute, specify the | |
Search CTEP, DCP, or All Trials | To narrow the search by this trial attribute, specify the sponsor organization. Otherwise, select All Trials. Valid values are as follows:
DCP is an abbreviation of Division of Cancer Prevention. CTEP is an abbreviation of Cancer Therapy Evaluation Program. PIO is an abbreviation of Protocol and Information Office. For information, refer to Abstracting Sponsors and Responsible Parties. |
Click Search, or press the Enter key on your keyboard.
The results of your query are listed in the Search Results section.
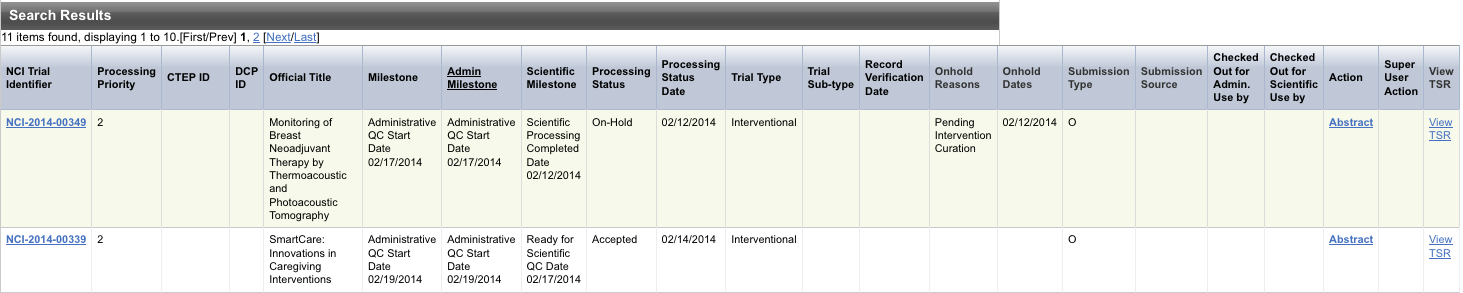
The system displays an asterisk (*) in the Official Title column for trials that have alternate titles.
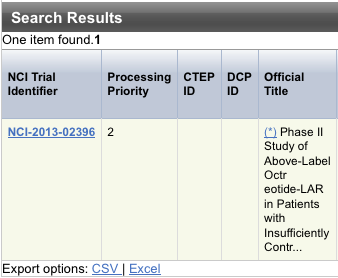
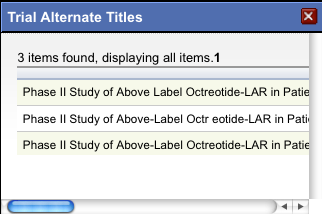
To view the alternate titles, click the asterisk.
Alternate titles are listed in the Trial Alternate Titles window.
To view details for a trial, click the NCI Trial Identifier link for the trial of interest, or click any link in the Action column.
The Trial Identification page displays trial identifiers and other details. It also displays one or more options to check out the trial for processing. For example, if you logged in to the application as a Super Abstractor, you can check the trial out for both administrative and scientific processing, as in the figure below. However, if you logged in as an Administrative Abstractor, your only option is to check in/out the trial for administrative processing.
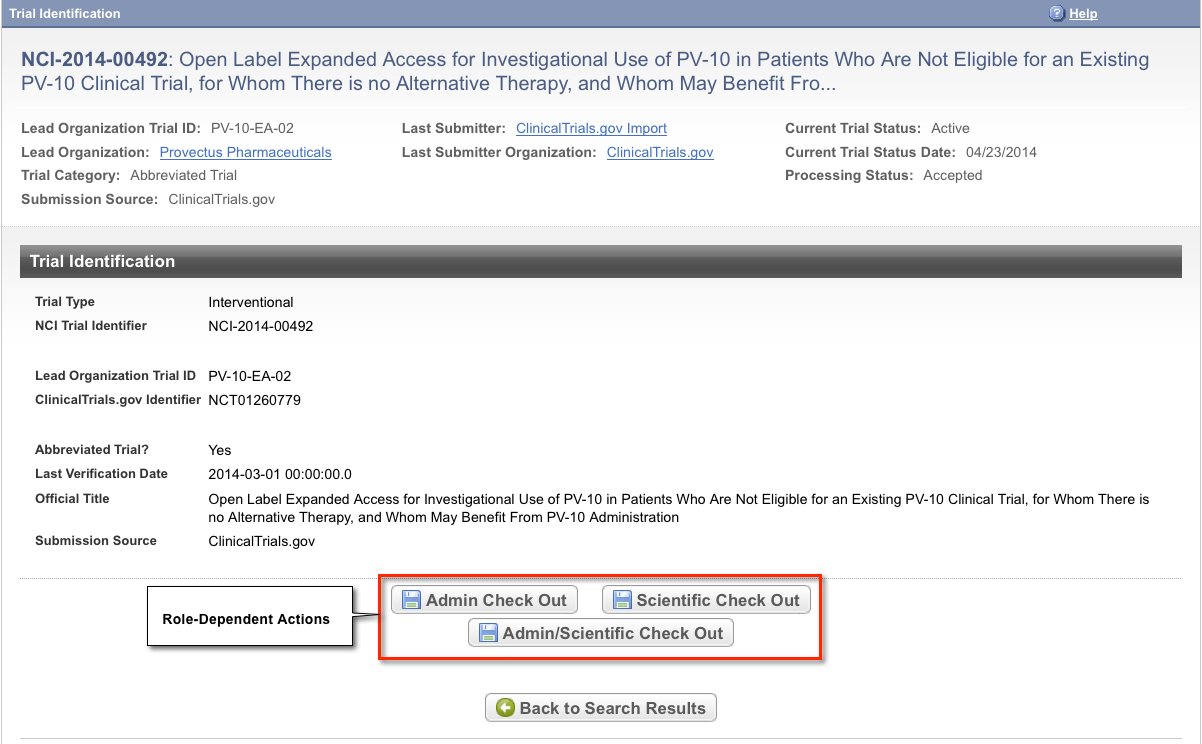
| To return to your most recent search results, click Back to Search Results on the Trial Identification page. |
- Click CSV to create a generic comma-separated value file.
- Click Excel to create a Microsoft Excel spreadsheet.