For example, you can grant ownership of one trial to three CTRP users within your organization. Or, you can grant one user ownership of three trials.
Additionally, you can tell the system to send notifications about the trial(s) via email to the owners you assign.
For Abbreviated trials, owners are responsible for updating their affiliated organization's information on trials in which their site is a participant, using the Update My Site action. Owners can update the following information:
- Organization family's program code
- Organization's local trial identifier
- Site principal investigator
- Site recruitment status and dates
Trial ownership requirements
In certain circumstances, you may not be able to change trial ownership using the Manage Trial Ownership functionality. For example, you may not be able to change trial ownership if the trial status is Complete rather than Active, and the lead organization is not the trial submitter's affiliated organization. (Refer to Recording Trial Statuses.) However, you can request trial ownership changes by contacting the CTRO at NCICTRO@mail.nih.gov. When requesting trial ownership changes, include the new owner's first name, last name, and email address. Ensure that the user has a valid CTRP account. For instructions, refer the user to Creating a CTRP Account.
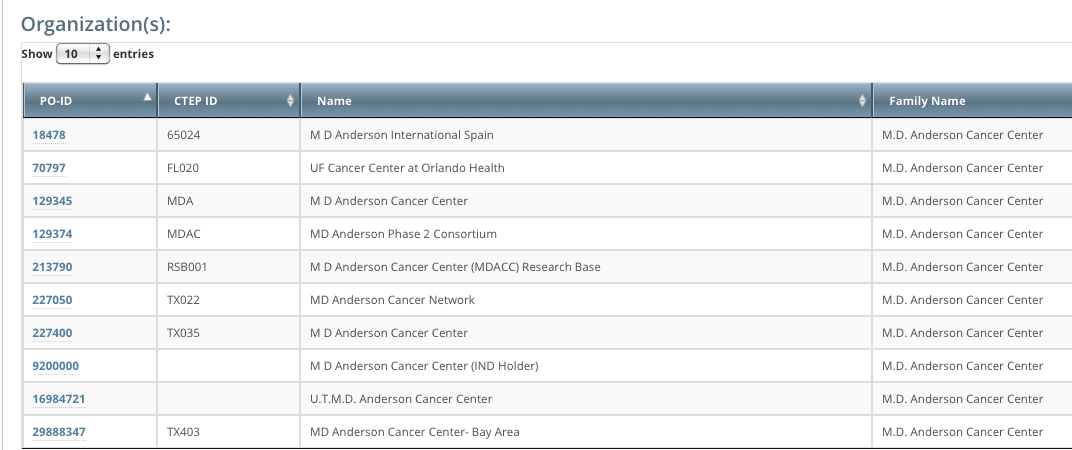
The system stores your affiliated organization with your account information on the My Account page. Your affiliation determines which trials and which users you can assign.
You can assign any trial submitted by your affiliated organization or its family member organizations. For example, if you are affiliated with the M D Anderson Cancer Center, you can manage trial assignments for trials associated with the M D Anderson International and UF Cancer Center at Orlando Health organizations. The following image lists the organizations in the M.D. Anderson Cancer Center organization family.
The organization must be a lead organization for Complete trials, or a participating site for Abbreviated trials.