Validate participating sites for Abbreviated trials.
Participating sites validation does not pertain to Complete trials.
Ensure that the following information is complete and accurate:
- Organization Name
- Local Trial Identifier
- Site Principal Investigator
- Site Recruitment Status
- Site Recruitment Status Date
- Date Opened for Accrual
- Date Closed for Accrual
How to Validate Participating Sites
- Select a trial to validate by following the instructions in Selecting Trials that Require Validation. The Trial Identification page displays the trial details.
- On the Trial Identification page, check out the trial. For instructions, see Checking In and Checking Out Trials.
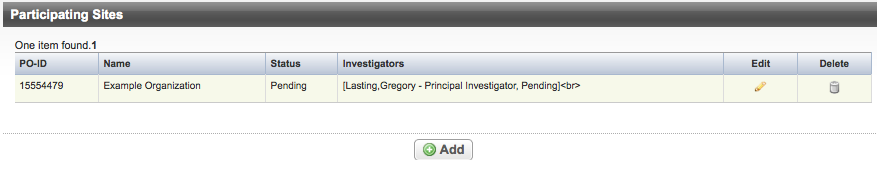
- On the Validation menu, click Participating Sites.
The Participating Sites Page appears.
- Do one of the following to continue:
- To add participating sites, click Add and follow the instructions provided in Abstracting Participating Sites.
- To modify an existing record, click the Edit icon and follow the instructions provided in Abstracting Participating Sites.
- To delete a record, click the Delete icon.
- If you made any changes, click Save.