You can retrieve existing trial records once you have registered for an account. For instructions, refer to Creating a CTRP Account.
After you have selected your search criteria, you can further limit or expand your search for trials as follows:
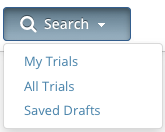
- Use the Search All Trials feature to search for all trials registered with the CTRP from all organizations/accounts, whether or not you are the submitter or owner.
- Use the Search My Trials feature to search for trials that you own, whether or not your organization is listed as the lead organization or participating site.
- Use the Search Saved Drafts feature to search for trials that you have saved as drafts but have not submitted.
The search feature you choose determines which categories of trials will be returned, and the actions you can perform with those results, as shown in the table below. For rules that determine which trial details are displayed, refer to Clinical Trial Search Results.
Search Option | Search All Trials | Search My Trials | Search Saved Drafts |
|---|---|---|---|
Types of Trials Returned | All trials |
| Partial Submissions |
Actions Permitted |
|
|
|
All registered users can search trials with the "Accepted" and subsequent processing status. Additionally, you can search for trials that you own that have not been validated. These trials are indicated by the "Submitted" status. See Trial Processing Statuses for information about statuses that occur during the course of the trial processing workflow.
How to Search for Existing Trials
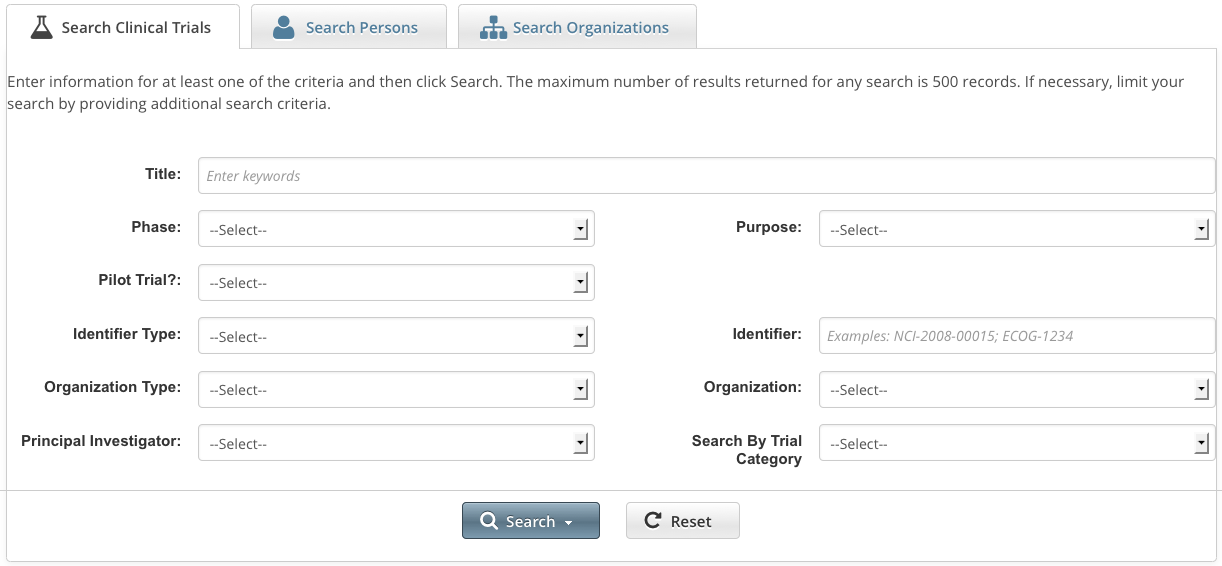
- Click the Search Clinical Trials tab. Or, on the toolbar, click Search > Clinical Trials.
The Search Clinical Trials page appears.
Select or enter the appropriate information in the drop-down lists and text fields. (You do not have to select or enter any search criteria if you use the Search My Trials feature. When searching All Trials, you must select or enter at least one search criterion.) The following table describes the fields.
Tip
If you are searching for a saved draft, you can search by the following criteria only:
- Title
- Phase
- Purpose
- Identifier Type (NCT Exact Match, Lead Organization, or Other Identifier only)
- Organization Type (Lead Organization only)
- Organization
- Principal Investigator
Because the system adds wildcards for you, do not enter wildcard symbols in the search fields.
Trial Search Criteria
To search by this...
Do this...
Title
Enter one or more words from the long title or name of the trial provided by the principal investigator or sponsor.
Avoid copying and pasting, or typing the entire title into the search field
Use keywords rather than phrases or the entire title. Doing so minimizes the potential for excluding from the search results any titles with misspellings or slightly different phrasing.
Phase
Select the trial phase from the drop-down menu. Valid values are as follows:
When you specify trial data in CTRP, the system requires you to specify the phase of investigation, as defined by the US FDA for trials involving investigational new drugs. The following table provides a definition of each phase and maps the ClinicalTrials.gov values to the CTRP values:CTRP Phase Value ClinicalTrials.gov Phase Value Definition Early Phase I Early Phase 1 (Formerly listed as "Phase 0") Exploratory trials, involving very limited human exposure, with no therapeutic or diagnostic intent (e.g., screening studies, microdose studies). See FDA guidance on exploratory IND studies for more information. I Phase 1 Includes initial studies to determine the metabolism and pharmacologic actions of drugs in humans, the side effects associated with increasing doses, and to gain early evidence of effectiveness; may include healthy participants and/or patients. I/II Phase 1/Phase 2 Trials that are a combination of phases 1 and 2. II Phase 2 Includes controlled clinical studies conducted to evaluate the effectiveness of the drug for a particular indication or indications in participants with the disease or condition under study and to determine the common short-term side effects and risks. II/III Phase 2/Phase 3 Trials that are a combination of phases 2 and 3. III Phase 3 Includes trials conducted after preliminary evidence suggesting effectiveness of the drug has been obtained, and are intended to gather additional information to evaluate the overall benefit-risk relationship of the drug. IV Phase 4 Studies of FDA-approved drugs to delineate additional information including the drug's risks, benefits, and optimal use. NA N/A Trials without phases (for example, studies of devices or behavioral interventions). Pilot Trial?
Select a value to narrow the search by this trial attribute:
- To find trials identified as pilot trials, select Yes.
- To find trials identified as not pilot trials, select No.
- To find trial records on which no pilot value has been specified, select None.
Purpose
Select the primary purpose of the trial from the drop-down list. Valid values are as follows:
CTRP allows you to choose one of the following values when specifying the main reason for conducting the trial.CTRP Primary Purpose Value Definition Treatment Protocol is designed to evaluate one or more interventions for treating a disease, syndrome, or condition. Prevention Protocol is designed to assess one or more interventions aimed at preventing the development of a specific disease or health condition. Supportive Care Protocol is designed to evaluate one or more interventions where the primary intent is to maximize comfort, minimize side effects or mitigate against a decline in the subject's health or function. In general, supportive care interventions are not intended to cure a disease. Screening Protocol is designed to assess or examine methods of identifying a condition (or risk factors for a condition) in people who are not yet known to have the condition (or risk factor). Diagnostic Protocol is designed to evaluate one or more interventions aimed at identifying a disease or health condition. Health Services Research Protocol is designed to evaluate the delivery, processes, management, organization, or financing of health care. Basic Science Protocol is designed to examine the basic mechanism of action (e.g., physiology, biomechanics) of an intervention. Device Feasibility An intervention of a device product is being evaluated in a small clinical trial (generally fewer than 10 participants) to determine the feasibility of the product; or a clinical trial to test a prototype device for feasibility and not health outcomes. Such studies are conducted to confirm the design and operating specifications of a device before beginning a full clinical trial.
Other Any purpose not described above. Identifier Type
Select the type of trial identifier from the drop-down list. Valid values are as follows:
- NCI - NIH National Cancer Institute identifier.
- NCT (Exact Match) - ClinicalTrials.gov identifier. Provide the exact number, including the NCT prefix. Example: NCT00012345
- Lead Organization - Enter the unique identifier assigned to the trial by the lead organization.
- Other identifier - Additional trial identifier such as unique identifier from other registries, NIH grant numbers, or protocol numbers assigned by the Review Board.
Selecting a Trial Identifier Type is not required
You can enter an identifier in the Identifier field without first having to choose an Identifier Type.
Identifier
Enter the unique identifier assigned to the trial by the NCI, ClinicalTrials.gov, PRS, or the identifier assigned to it by the lead organization. For Inter-Group trials, enter the Lead Group's trial number.
Organization Type
Select one of the following organization roles from the drop-down list:
- Lead Organization - Returns all trials on which the selected organization is the Lead Organization
- Participating Site - Returns all trials on which the selected organization is a Participating Site
Both - Returns all trials on which the selected organization is either the Lead Organization or Participating Site
You can change the Organization Type without affecting any other search criteria you may have selected previously.
Organization
The system suggests organizations as you type.
Enter the initial letter(s) of the organization and then select the organization from the list of suggestions.
To search for trials by organization without having to specify what role the organization plays in the trial, select Both from the Organization Type list, and then select the name of the organization of interest.
Principal Investigator
The system suggests names as you type.
Enter the initial letter(s) of the principal investigator's last name and then select the investigator name from the list of suggestions.
Search by Trial Category
To restrict your search by trial category, select Abbreviated or Complete from the drop-down list. For information about these categories, refer to CTRP Trial Categories (Study Sources). Otherwise, select Both.
Click Search.
The Search menu options are displayed.- Do one of the following:
- To search all registered trials in the system, click All Trials.
-or- To search only the trials that you submitted or own, click My Trials. This feature enables access to all the trials that you have submitted, including those that are currently on hold. (The Clinical Trials Reporting Office staff places trials on hold when they are unable to process a trial without further information, usually from the submitter.)
-or-
- To search only the trials that you have saved for later completion, click Saved Drafts.
-or- To clear all search criteria and begin a new search, click Reset.
Allow sufficient time for the system to conduct your search before you run your search again
The search is complete only when the system displays search results or alerts you that it could not find a trial to match your search criteria.
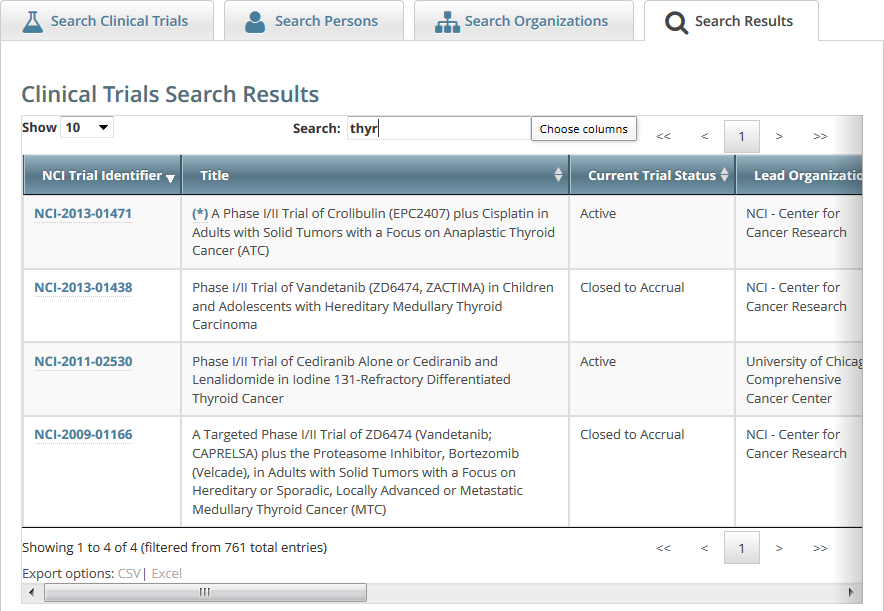
The trials that meet your search criteria are listed on the Search Results page. For more information on navigating and working with search results, see Working with Tables and Search Results.
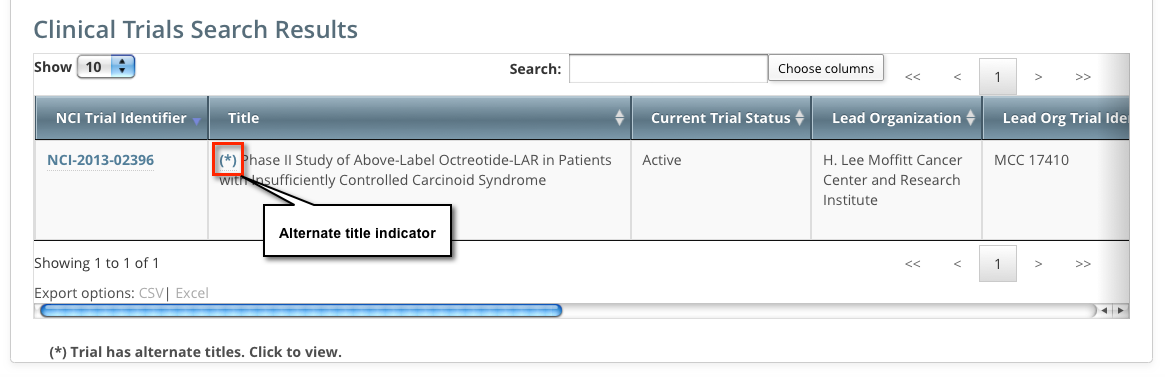
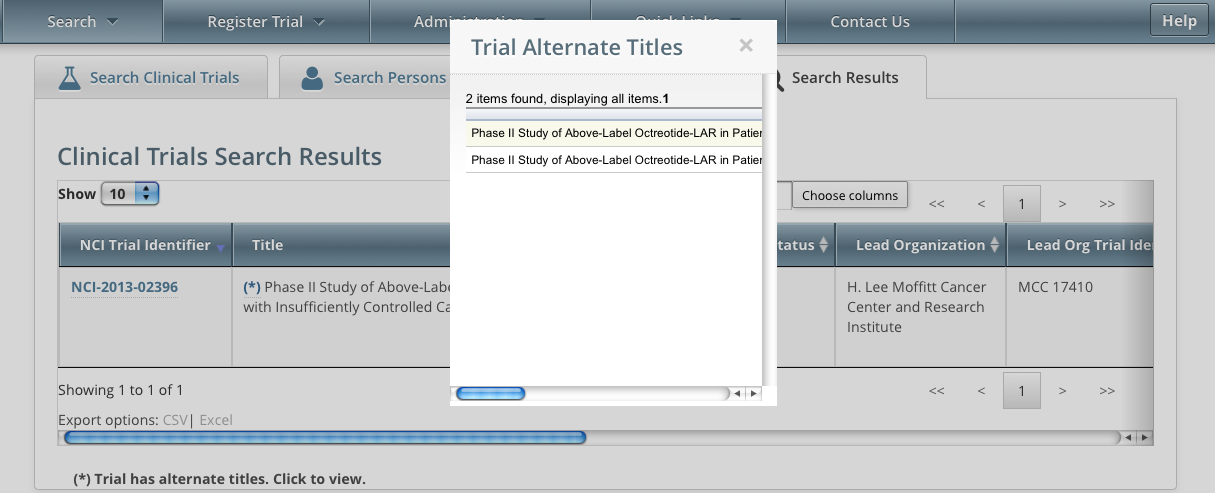
Trials may have more than one title. For example, the CTRO staffs may add an alternate title if they find a misspelling in the registered title. Any trial identified by more than one title is identified in the search results table by an asterisk ( * ) in the Title column.
- To search all registered trials in the system, click All Trials.
To see the alternate titles associated with a trial, click the asterisk (link).
The list of alternate titles is displayed in the Trial Alternate Titles window.
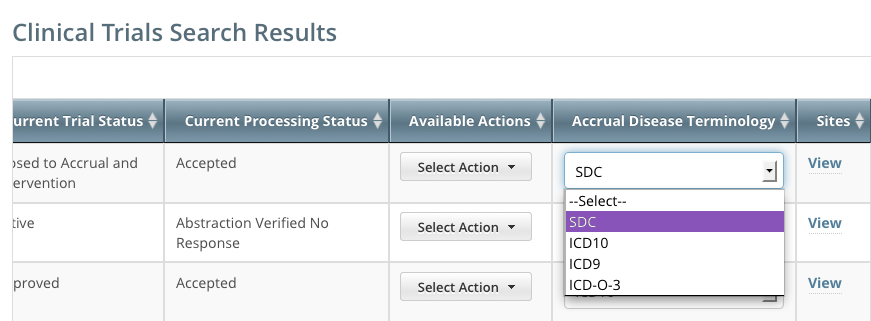
You can change Accrual Disease terminologies for individual trials
If you searched for "My Trials", the search results table displays an additional column, Accrual Disease Terminology. You can select a new terminology from the drop-down list only if the trial has not accrued patients.
Additionally, you can change accrual disease terminology at any time for trials currently recording accruals at the summary level only.
- To view a trial, click its NCI Trial Identifier link. The Trial Details page appears. For information about this page, refer to Viewing Trial Details.
For more information, refer to the following pages: