Complete all fields in the Data Table 4 Information section. The Trial Submission Category (study source) or Data Table 4 Funding Sponsor Type fields are pre-populated with the sponsor type.
The lead organization or at least one participating site must be a Cancer Center organization. |
How to Complete the Data Table 4 Information Section
Select the name of the external sponsor or funding source as defined by the Data Table 4 report. Refer to Looking Up Registered Organizations. (If your trial's funding sponsor is not listed, you can ask the CTRO to create the organization record. To do so, follow the instructions in Requesting Creation of an Organization Record.) 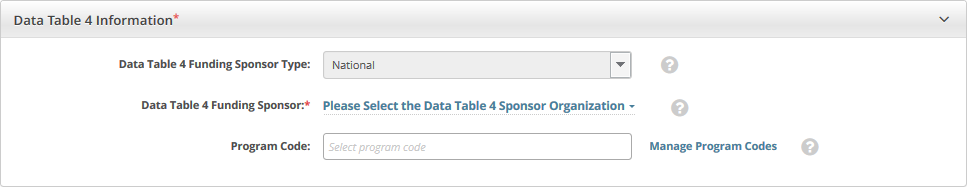
The organization you selected appears under the Data Table 4 Funding Sponsor field, along with an option to delete the sponsor.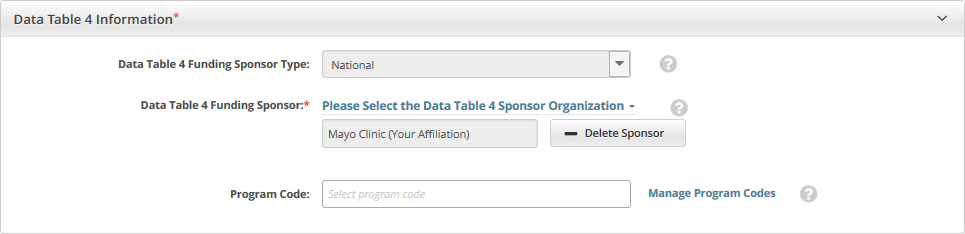
A trial can have multiple sponsors. Repeat the steps above to add other sponsors. The system ensures that you don't duplicate an existing sponsor. |
To delete an existing sponsor, click Delete Sponsor. You can not "undo" the deletion but you can add the sponsor back if necessary.
If you have selected a lead organization for the trial and if that lead organization belongs to an organization family, the Program Code field is available. The Program Code field lists all program codes available for that organization family. If you are a site administrator, an option to manage program codes is also available.